Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Sugar Picker
Synthetic receptor selects cellobiose out of a sugary mix
by Bethany Halford
October 29, 2007
| A version of this story appeared in
Volume 85, Issue 44
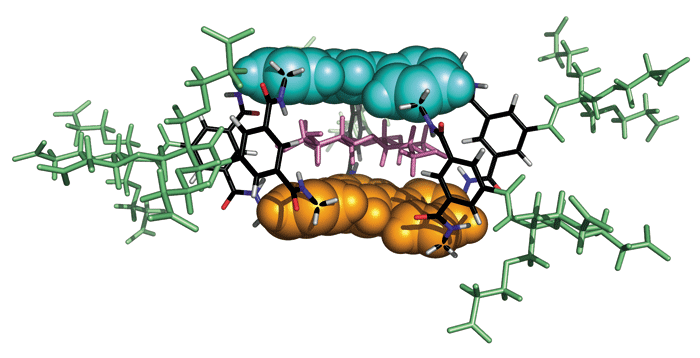
LECTINS ARE MASTERS of recognition. These carbohydrate-binding proteins can suss out saccharides, distinguishing their highly hydroxylated structures from a sea of water molecules. Now, chemists in England have created a synthetic lectin capable of selectively binding to a specific disaccharide in aqueous solution (Science 2007, 318, 619).
"We made the lectin analog to prove that you can use synthetic molecules to mimic the behavior of proteins," says Anthony P. Davis, a professor of supramolecular chemistry at the University of Bristol who spearheaded the project.
Davis and coworkers Yann Ferrand and Matthew P. Crump designed their receptor to selectively bind cellobiose, a disaccharide that has only hydrogen atoms in its axial positions. The rest of the sugar's substituents occupy equatorial positions.
The synthetic lectin has a hydrophobic roof and floor made up of meta-terphenyl groups, which interact with the cellobiose's axial C-H groups. Five isophthalamide pillars define the length of the cavity where the disaccharide binds. The isophthalamide's amide groups hydrogen-bond to the hydroxyl groups of the sugar substrate. Tricarboxylate moieties attached to each isophthalamide make the receptor water soluble.
The researchers found that the synthetic receptor displays a good affinity for cellobiose, matching the strength of some lectin-carbohydrate interactions. The lectin analog proved to be remarkably selective, preferentially binding to cellobiose over similar disaccharides such as lactose.
Geert-Jan Boons, a chemistry professor and carbohydrate expert at the University of Georgia, says Davis' cleverly designed lectin analog is an important step toward developing synthetic carbohydrate receptors. "The results provide strong encouragement for the design of receptors that can recognize biologically interesting saccharides linked to glycoproteins and glycolipids," he says.
Davis tells C&EN that his group hopes to use the information they've gleaned from the cellobiose receptor to make a lectin analog that specifically binds glucose and could be used as a glucose sensor.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter