Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Cancer Stem Cells
Researchers zero in on the pathways that allow cancer to bounce back after treatment
by Aaron A. Rowe
October 24, 2011
| A version of this story appeared in
Volume 89, Issue 43

Take some cells from a tough-to-treat tumor, sort them, and inject each fraction into a different immunodeficient mouse, and only a small percentage of those cells will thrive and form tumors.
This sort of experiment illustrates a concept that has been gaining traction within the cancer research community. Tumors contain a diverse mixture of cells, and only a handful of them can bounce back after treatment. That deadly minority can reproduce indefinitely and differentiate into a wide variety of cell types, just like stem cells. And often they express many of the same genes that are active in induced or embryonic stem cells and inactive in mature tissue.
Not all cancers fit seamlessly into this paradigm. Until recently, the hypothesis that stemlike cells are at the root of cancer recurrence was mired in controversy. But in the past few years, a great deal of evidence has shown that it holds true in many types of leukemia, breast cancer, and brain cancer. In these diseases, the proportion of cancer cells with a stemlike character can sometimes be as high as 40%. On the other hand, some cancers don’t contain any cells that exhibit stem cell behavior.
Many of today’s cancer treatments can wipe out the most vulnerable cells in a tumor, but they often leave behind cells with tremendous regenerative ability. It’s like mowing down a field of dandelions but leaving their roots intact, says William Matsui, an oncologist from Johns Hopkins University School of Medicine who studies cancer stem cells. He explains that measuring the shrinkage of tumors is an awful way to gauge the effectiveness of a cancer treatment. Statistics from large-scale studies of cancer patients show little correlation between how much their tumors shrink and how long they live. Instead, Matsui argues, cancer treatments should be evaluated for the ability to prevent relapses.
Many scientists are becoming convinced that targeting cancer stem cells is the key to preventing relapses. But finding drugs with a robust effect on cancer stem cells isn’t easy. Some stem cells are inherently drug resistant because they replicate slowly compared with mature cancer cells and express proteins that pump out or otherwise eliminate small molecules. Even worse, some stem cells can continue replicating even after traditional anticancer drugs shut down their primary cancer-causing mutation.
Despite these obstacles, many cancer research groups have begun to hunt for molecules that target stem cell pathways or developmental pathways that influence the fate of stemlike cells in tumors. They are approaching the problem with many classes of therapeutics, from small RNAs and hormones to natural products.
So far, protein-based strategies have shown the most promise. One example is a protein called granulocyte colony-stimulating factor. G-CSF can force stem cells to differentiate into mature cells. It’s often used to increase the effectiveness of bone marrow or cord blood transplants. In 2003, a team of European researchers showed that G-CSF can also increase the disease-free survival times of patients with acute myeloid leukemia (New Eng. J. Med., DOI: 10.1056/NEJMoa025406). It wipes out the leukemia stem cells by forcing them to become granulocytes and other mature cells.
Antibodies also show promise as a means of eradicating cancer stem cells. In 2003, Michael F. Clarke and colleagues at Stanford University showed that breast cancer stem cells can be identified by their expression of CD44, a well-known cell-surface marker. They founded a company called OncoMed and have advanced two antibodies (OMP-21M18 and OMP-59R5) into the clinic. More recently, ImmunoCellular Therapeutics, a Los Angeles-based cancer vaccine start-up, has been developing cancer vaccines that target the stem cell subpopulation in tumors. They reported that their flagship product increased long-term survival in 55% of glioblastoma patients who enrolled in a Phase I trial.
The hunt for small molecules that exclusively target cancer stem cells remains at an even earlier stage, according to Waldemar Priebe, a professor of medicinal chemistry at the University of Texas M. D. Anderson Cancer Center. His group recently identified and modified a natural product that inhibits STAT3, a transcription factor that is upregulated in both cancer cells and embryonic stem cells. The modified natural product, dubbed WP1066, halts the growth of both normal and stemlike cancer cells, while preventing them from secreting molecules that suppress the immune system.
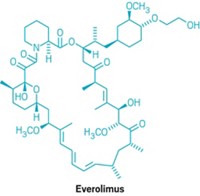
Hoping to expedite the small-molecule hunt, many cancer researchers have tested Food & Drug Administration-approved drugs for their effect on cancer stem cells. In animal experiments, the immunosuppressant rapamycin has shown promise in defeating neuroblastoma stem cells and the diabetes drug metformin helped doxorubicin wipe out breast cancer stem cells.
Meanwhile, a handful of labs are testing new substances that act on well-studied developmental pathways. Blocking developmental signals can interfere with cancer stem cells’ ability to reproduce, according to Margaret A. Read, a senior product developer at Infinity Pharmaceuticals.
The company has already found one such compound. Infinity’s IPI-926 inhibits Smoothened, a regulator of the hedgehog signaling pathway that is well-known for its role in developmental biology. IPI-926 has completed Phase I trials and is being evaluated in several other Phase I and II trials for indications including pancreatic cancer, squamous cell carcinoma, and chondrosarcoma.
A few academic research groups have started looking for drug targets within developmental pathways that aren’t nearly as famous as hedgehog. For instance, at the University of California, San Diego, Tannishtha Reya and her group have found that disrupting the Musashi-Numb signaling axis could knock out the stem cells behind chronic myelogenous leukemia (Nature, DOI: 10.1038/nature09171). By blocking the production of the Musashi protein with short hairpin-shaped RNA molecules, they prevented leukemia cells from adopting a particularly aggressive and deadly phenotype. Her team is now beginning to screen small-molecule inhibitors of this pathway to find leukemia drug leads.
To further accelerate the drug hunt, some researchers are looking for cancer stem cell stand-ins that might prove viable test beds for screening new cancer therapies. At the Whitehead Institute, Robert A. Weinberg and his collaborators have found ways to generate a large number of mesenchymal stem cells. Derived from epithelial cells, mesenchymal cells are thought to be potential precursors to cancer stem cells. His team has used these cells to screen potential anticancer drugs. They’ve identified several lead compounds, including salinomycin, which destroys cancer stem cells 100 times more efficiently than paclitaxel does (Cell, DOI: 10.1016/j.cell.2009.06.034). Weinberg and colleagues Piyush B. Gupta and Eric S. Lander have cofounded a company, Verastem, to pursue these leads.
Another group has begun screening compounds on cultures with an enriched population of cancer stem cells. Harley Kornblum and colleagues at UCLA, recently screened 31,624 compounds and narrowed the pool down to four leads. In addition to looking at the effect of the compounds on cell growth, they analyzed the effects on the expression of four genes that are correlated with a poor clinical outcome for cancer patients (Mol. Cancer Ther., DOI: 10.1158/1535-7163.MCT-11-0268). One of the four leads discovered in their study was emetine, an antiprotozoal drug.
RNA interference has proven itself a useful tool for validating targets within cancer stem cells. It has been particularly useful in targeting the same transcription factors that are used to generate induced pluripotent stem cells. Many in the cancer stem cell field think these transcription factors could become important cancer drug targets.
For example, researchers who study induced pluripotent stem cells quickly learned that c-Myc, one of the transcription factors, causes the cells to become cancerous if c-Myc is left active indefinitely. So they’ve learned to only transiently turn it on. Using hairpin-shaped short RNA molecules to knock down c-Myc, Jeremy Rich of the Cleveland Clinic and colleagues from Duke University showed that it is required for the upkeep of glioma stem cells (PLoS One, DOI: 10.1371/journal.pone.0003769). Another group of researchers, led by Ivan Stamenkovic of the University of Lausanne in Switzerland, showed that three more of these transcription factors, Oct4, SOX2, and Nanog, are active in Ewing’s sarcoma (Genes Dev., DOI: 10.1101/gad.1899710).
The logic of pursuing therapies that might zero in on cancer stem cells is compelling to many. But the methods to evaluate such therapies’ effectiveness, or to personalize cancer treatments according to stem cell markers, are not nearly as well developed. Without an array of proper markers, it’s hard to tell whether drugs that target cancer stem cells are working as intended.
Only recently, genetic markers of cancer stem cells have started showing promise. Some of the most rigorous work in this field was done by John E. Dick of the University of Toronto and colleagues. Using blood samples from 16 patients with acute myeloid leukemia, they identified a gene expression signature that signals the aberrant behavior characteristic of cancer stem cells and a poor prognosis for patients (Nat. Med., DOI: 10.1038/nm.2415). Similarly, Stephen R. Quake, Irving L. Weissman, and colleagues at Stanford University are developing methods to measure gene expression in single stem cells (Nat. Biotechnol., DOI: 10.1038/nbt.1977). This sort of analysis will be particularly useful for studying cancer stem cells, because they are in the minority and the gene expression patterns can easily be masked when cancer cells are analyzed in bulk, rather than individually.
Things are looking up for genetic analysis, but the poor reliability of cancer stem-cell-surface markers remains a confounding problem. For nearly a decade, biologists have known that antigens such as CD133 can be found on the surfaces of cancer stem cells. But these markers are not particularly specific. It would be nice if the newest technologies for monitoring circulating tumor cells via surface markers could be adapted to monitor cancer stem cell populations during clinical trials, says Inifinity’s Read.
But for solid tumors, which account for about 85% of all cancer diagnoses, the search for such stem-cell-surface markers is still in the early days. In such cancers, Read notes, cell-surface markers can vary from one type of cancer to another or even from one cell within a tumor to another. Until better markers are discovered, she adds, the cancer stem cell field will remain somewhat embryonic. ◾







Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter