Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Catalysis Merger Leads to Mild Arylation
Combination of photocatalysis with palladium catalysis could be broadly applicable
by Carmen Drahl
November 7, 2011
| A version of this story appeared in
Volume 89, Issue 45

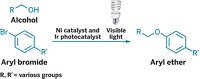
By marrying two catalytic cycles—one mediated by palladium and the other by light—chemists at the University of Michigan have developed a C–H arylation reaction that proceeds at room temperature (J. Am. Chem. Soc., DOI: 10.1021/ja208068w). The merger is “a far-reaching concept” that could extend the capabilities of C–H activation chemistry for making pharmaceuticals and materials, says chemistry professor Melanie S. Sanford, who led the Michigan team.
Aryl-aryl bonds are common motifs, but C–H activation routes to these bonds require acidic solvents, elevated temperatures, or both. To improve that state of affairs, postdoc Dipannita Kalyani and graduate students Kate B. McMurtrey and Sharon R. Neufeldt revisited a C–H arylation reaction Sanford developed in 2005. That reaction barely worked at room temperature. But after studying the reaction’s mechanism, the team realized they could fix that by changing their arylating reagent to something more reactive—aryl radicals.
To generate aryl radicals under mild conditions, the Michigan team looked to an emerging movement—combining catalytic processes (C&EN, Sept. 8, 2008, page 10). By adding a ruthenium(II) bipyridine catalyst to their palladium-catalyzed process, Sanford’s team harvested light from a compact fluorescent bulb to produce aryl radicals. The room-temperature method is compatible with functional groups, such as oximes, that don’t behave well in traditional C–H activations.
In addition to studying the reaction’s mechanism, Sanford’s team is exploring ways to extend the merger concept. “All sorts of radicals and reactive species can be generated with photocatalysis,” she says. Coupling those species with palladium-catalyzed C–C bond formations would open up many new synthetic doors, she adds.
“I am convinced that the mild nature of this transformation renders it very attractive for sophisticated applications, including C–H activations of sensitive materials like metal-organic frameworks,” says Frank Glorius, who studies C–H activation at the University of Münster, in Germany. “Moreover, I expect this work to inspire many more related processes using other catalytically active metals.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter