Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Neuropeptide Signaling Seen In A Different Light
Photoactivated neuropeptides enable researchers to study kinetics of receptor signaling in the brain
by Lauren K. Wolf
February 6, 2012
| A version of this story appeared in
Volume 90, Issue 6
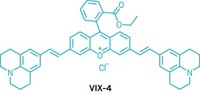
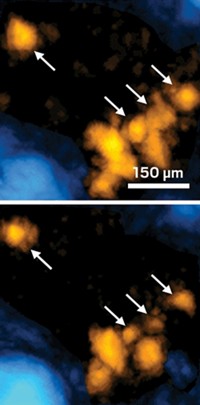
Researchers at Harvard Medical School have designed a light-activated neuropeptide for studying signaling between neurons in the brain (Neuron, DOI: 10.1016/j.neuron.2011.11.016). Like small-molecule neurotransmitters, the larger neuropeptides trigger protein receptors and alter neuron firing. But the peptides diffuse more slowly and travel over larger regions than neurotransmitters, making them difficult to study. Matthew R. Banghart and Bernardo L. Sabatini have now developed a photoactivatable version of the neuropeptide [Leu5]-enkephalin, an opioid. By virtue of a carboxynitrobenzyl protecting group (shown in red) that can be cleaved from the peptide with ultraviolet light, the scientists are able to precisely control where, when, and how much of the active peptide gets released in brain tissue. After equilibrating the protected neuropeptide in rat brain slices, they activated the enkephalin with a laser beam and, for the first time, measured how fast it triggered opioid receptors in nearby neurons. In doing so, Banghart and Sabatini also determined that the neuropeptide can travel as far as 300 µm from its release point and still activate nerve receptors. This is remarkable, Banghart says, given that small-molecule neurotransmitters travel only a few micrometers after being released from nerve cells in synapses.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter