Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Altered Antibodies Offer Improved Conjugates
Developers use mutagenesis to create specific sites for conjugating drugs
by Ann M. Thayer
January 20, 2014
| A version of this story appeared in
Volume 92, Issue 3
Most antibody-drug conjugates in clinical development have been made by attaching drug molecules to available sites in an antibody’s natural structure. Although successful, this approach can lead to inhomogeneous products. Recent developments around ADCs center on creating well-defined structures with a set number of drug molecules in known locations. The belief is that doing so will not only make ADCs easier to purify and characterize but also more stable and functional.
COVER STORY
Altered Antibodies Offer Improved Conjugates
Through small changes to an antibody’s amino acid sequence, specific sites can be constructed for conjugating drug payloads. The catch is that the approach requires antibodies to be engineered with this in mind—or reengineered if a previously developed antibody is used. Companies offering site-specific conjugation technology generally say that such changes are simple enough to make and worth the improved ADC quality and performance.
Moving away from the natural antibody, the California-based ADC developers Sutro Biopharma and Ambrx have separately chosen to incorporate nonnatural amino acids into antibodies to create sites for drug conjugation. Their ADC work has attracted major pharma partners: Ambrx is collaborating with Astellas Pharma, Bristol-Myers Squibb, and Merck & Co.; Sutro is working with Celgene.
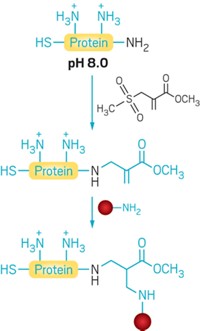
Meanwhile, the approach of California-based Redwood Bioscience with its SMARTag technology is to place a chemical handle at predetermined locations in an antibody by inserting an all-natural five-amino-acid sequence. A formylglycine-generating enzyme recognizes the sequence tag and selectively converts it into a unique aldehyde-bearing amino acid. At this engineered site, the aldehyde is reacted using aldehyde-selective chemistries to join linkers and drugs.
Conjugation is site specific and very stable, says Redwood Chief Scientific Officer David Rabuka. “We never see conjugation happening anywhere else on the protein.” And to determine the best sites for making an ADC, “we have mapped all around the antibody where we would like to put tags and have generated a very large number of different antibodies to explore the effect.” Since April, Redwood has been working with Catalent Pharma Solutions to scale its technology.
In October, the French company Innate Pharma described a new conjugation technology that uses a bacterial transglutaminase enzyme. By introducing single-point mutations in an antibody, Innate can generate enzyme-recognition sites, usually two or four. The enzyme then attaches a drug-carrying linker the company has designed to couple specifically at these sites to create ADCs with drug-to-antibody ratios (DARs) of two or four.
Relying both on what antibodies have to offer and some genetic engineering, Meditope Biosciences is using technology developed in the lab of John C. Williams, an associate professor at City of Hope, a cancer center in Duarte, Calif. He found an antibody that contains a specific short amino acid sequence within the two arms of its Y shape. He also discovered a specific peptide, dubbed a “meditope,” that attaches there. The site is far enough away from the antibody’s target-binding region on the ends of the arms that using it doesn’t interfere with the antibody’s desired properties.
By inserting the short amino acid sequence, any antibody can be “meditope-enabled,” Williams explains. Likewise, the meditope itself can be easily synthesized and conjugated to drug molecules. When the two parts are simply mixed—no chemical conjugation is needed—the drug-carrying meditope joins noncovalently, but with high affinity, to the modified antibody.
The result is an ADC with a known and consistent DAR. And its stability looks promising, despite the lack of chemical bonding. Flexibility may be another feature. For a given meditope-enabled antibody, a range of possible ADCs can be designed and tested by creating meditopes with different drug molecules.
“Once you have the hitch on, you can put any trailer you want to on the truck,” Williams says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter