Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Ebola Prevention: Researchers Get Ready To Test Two Vaccines In West Africa
by Bethany Halford
November 26, 2014
| A version of this story appeared in
Volume 92, Issue 48
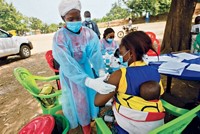
While many scientists are hard at work to find therapies to treat Ebola patients, other researchers are concentrating their efforts on creating vaccines to keep the virus from infecting people in the first place.
COVER STORY
Ebola Prevention: Researchers Get Ready To Test Two Vaccines In West Africa
Scientists have searched for an Ebola vaccine since the first outbreak of the disease in 1976. Injections of inactivated virus proved to be ineffective in nonhuman primates. Over the intervening decades a number of classical vaccine approaches were tried, but none proved effective, and funding for such research has been scarce.
In recent months, two different Ebola vaccines—ChAd3 and VSV-EBOV—have undergone Phase I trials in healthy human volunteers to determine whether they are safe and what their appropriate doses are. Larger trials of the vaccines’ efficacy could begin as early as this month in Sierra Leone and Liberia, two Ebola-stricken countries in West Africa. People expected to get the experimental vaccines as part of the larger trial include health care workers.
Scientists at the National Institute of Allergy & Infectious Diseases (NIAID) originally developed ChAd3 in collaboration with the biotech company Okairos. GlaxoSmithKline (GSK) bought Okairos and its Ebola vaccine last year.
ChAd3 is made from an adenovirus that affects chimpanzees. The adenovirus acts as a carrier, delivering the gene that makes the Ebola glycoprotein, which decorates the surface of Ebola virus particles and is critical to infection. The modified adenovirus prompts the vaccine recipient’s cells to express the glycoprotein; subsequently, the body mounts an immune response to it. Because the adenovirus does not replicate, researchers anticipate that the ChAd3 vaccine will require a booster.
VSV-EBOV, a vaccine developed by the Public Health Agency of Canada and licensed last week by Merck & Co. from NewLink Genetics, does replicate, so researchers believe it can work with just one jab. It uses vesicular stomatitis virus, or VSV, a virus that affects livestock. Scientists swap VSV’s glycoprotein gene with the gene for making the Ebola glycoprotein. As with ChAd3, the vaccine recipient starts making the Ebola glycoprotein and then develops an immune response to it.
Even with large-scale trials starting for these two vaccines, doctors and scientists face plenty of challenges. Andrea Marzi, a virologist who studies Ebola at NIAID, notes that in West Africa, where access to low-temperature storage is limited, it will be tough to keep these vaccines at –80 °C, a temperature required to sustain activity.
Manufacturing presents another hurdle. “One challenge is finding sufficient sterile capacity for filling and packaging finished vials,” GSK spokeswoman Mary Anne Rhyne says. “Because Ebola vaccines use genetically modified organisms, the sterile filling of vials must be done in a biosafety level 2 facility, which only a few companies possess.”
Because of the crisis, Rhyne adds, GSK is taking the unusual step of ramping up production before it even knows if its vaccine is safe and effective.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter