Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
A Challenging Conjugation
Bioactivity: Linking to antibody fragment enhances immune-system-regulating agent
by Stu Borman
February 28, 2014
| A version of this story appeared in
Volume 92, Issue 9

By accomplishing a particularly difficult antibody conjugation, researchers have enhanced the therapeutic properties of a small-molecule immune-system-regulating agent. The technique could be applicable to other small molecules as well.
The small molecule CGS-21680 targets adenosine 2A receptors on immune system cells and is thus a potential treatment for immune system malfunction. But it breaks down quickly and can cause side effects by entering non-immune-system cells.
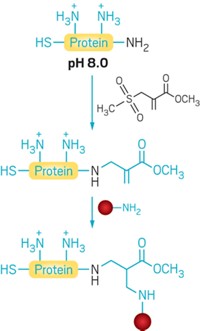
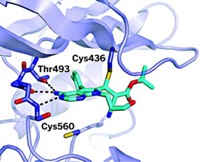
To address those problems, pharmacologists Jonathan D. Powell and Philip A. Cole of Johns Hopkins University School of Medicine and coworkers used a technique called expressed protein ligation (EPL) to conjugate the agent to an antibody fragment called an Fc domain (J. Am. Chem. Soc. 2014, DOI: 10.1021/ja5006674).
The antibody fragment resists degradation and localizes only to surfaces of immune system cells. When the conjugate was used to treat an autoimmune lung disease in mice, it was more potent, stable, and target-selective than CGS-21680, and it improved mouse survival.
Protein conjugation has been used before, but Cole notes that this is the first use of EPL for Fc-domain conjugation. The domain’s glycosylation and disulfides make site-specific conjugation difficult.
EPL, developed in 1998 by Cole and others, uses a protein fragment called an intein to form a thioester, in this case on the Fc domain. The thioester reacts with a thiol, on CGS-21680 in this example, to effect conjugation. EPL offers better site specificity, higher yields, and greater simplicity than other protein conjugation methods, the researchers note.
Pharmacologist David A. Scheinberg of Sloan Kettering Institute comments that the approach “has the potential to dramatically change the pharmacokinetics”—the absorption, distribution, metabolism, and excretion properties—“of a variety of small-molecule drugs.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter