Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
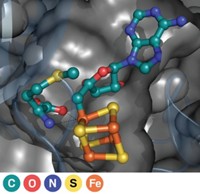
Enzyme’s Elusive Proton Donor Found
Enzyme Mechanisms: Active-site tyrosine is found to transfer the needed proton in enoyl-thioester reductase reactions
by Stu Borman
April 20, 2015
| A version of this story appeared in
Volume 93, Issue 16
Researchers have identified the long-sought proton donor in enoyl-thioester reductase (ETR) enzymes, enabling them to reengineer the stereochemistry of the enzymes’ catalytic action. The work has implications for polyketide synthase-based synthesis of natural products and natural-product-like compounds because related reductases are major components of polyketide synthase enzyme complexes. In reactions catalyzed by ETRs, a proton from an active-site amino acid or water molecule is transferred to the α-carbon of the enoyl-thioester substrate. But scientists haven’t been able to pin down the identity of the proton donor. Tobias J. Erb of the Max Planck Institute for Terrestrial Microbiology, in Marburg, Germany, and coworkers provided experimental evidence for an earlier proposal that an intermediate called a C2-ene adduct forms between the substrate and NADPH in the reaction, suggesting it might be involved in the proton-transfer process. Erb and coworkers’ use of this C2-ene adduct as a molecular probe of the reaction has now enabled them to identify an active-site tyrosine as the elusive proton donor (Nat. Chem. Biol. 2015, DOI: 10.1038/nchembio.1794). This new understanding also enabled them to reengineer the enzyme to catalyze the reaction with inverted stereochemistry.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter