Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Lilly Pulls Heart Drug
Pharmaceuticals: Development of CETP inhibitor evacetrapib fails in large Phase III study
by Lisa M. Jarvis
October 19, 2015
| A version of this story appeared in
Volume 93, Issue 41
In a blow to its late-stage drug pipeline, Eli Lilly & Co. is pulling the plug on evacetrapib after the cholesterol drug failed to improve cardiovascular outcomes in a huge patient study. In addition to sending Lilly’s stock down by nearly 9% last week, the setback shook the larger cardiovascular drug development field as watchers questioned the drug’s mechanism of action.
Evacetrapib is part of a class of compounds that raise levels of high-density lipoprotein—or “good” cholesterol—by blocking cholesteryl ester transfer protein (CETP). The strategy differs from statin drugs, which decrease levels of low-density lipoprotein—the “bad” cholesterol. In both cases the goal is to avoid heart attacks and strokes by reducing arterial plaque.
But early data from a Phase III clinical trial that enrolled more than 12,000 people suggested evacetrapib was not effective in reducing cardiovascular events. Lilly will take a $90 million charge in its fourth-quarter earnings as it shuts down the program. Had it been successful, evacetrapib was expected to hit the market in 2018 and rack up annual sales of as much as $4 billion. Now, analysts are questioning the health of Lilly’s new drug pipeline.
Lilly isn’t the only company to have been done wrong by the CETP target. In one of the most notable failures in the drug industry’s history, Pfizer shelved its CETP inhibitor torcetrapib in 2006 after it increased, rather than decreased, the risk of death in a 15,000-person study. Later studies revealed that the drug also failed to slow down the accumulation of plaque in heart arteries.
After the Pfizer bombshell, companies developing CETP inhibitors incorporated more safety and mechanistic studies into their programs. Merck & Co., Roche, and Lilly plowed ahead with CETP inhibitors that they said overcame the safety liability in Pfizer’s compound.
But Roche ended development of its CETP inhibitor, dalcetrapib, in 2012 because of lack of efficacy. With Lilly now reporting a safe, but ineffective, molecule, many are questioning whether the approach is flawed.
“The commercial prospect of the entire CETP inhibitor class is again in doubt, with three out of four major CETP outcome trials having failed,” Leerink stock analyst Seamus Fernandez said in a note to investors.
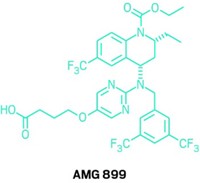
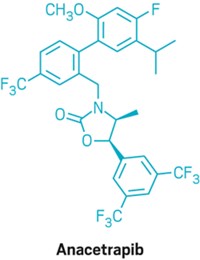
Merck continues to push forward with anacetrapib, which is currently being tested in a Phase III study that has enrolled more than 30,000 people. Amgen, meanwhile, paid $300 million last month to buy Dezima Pharma, which has a CETP inhibitor in Phase IIb studies.
John LaMattina, a senior partner at the health care venture firm PureTech Ventures, was head of R&D at Pfizer during the torcetrapib setback. He suggests Amgen should scrap its program rather than risk a hefty investment in large studies that seem destined to fail.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter