Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
New analytical method has a taste for catching counterfeit whiskeys
‘Artificial tongue’ can distinguish between high-value brands and cheap imitations of the malted beverage
by Sarah Everts
June 12, 2017
| A version of this story appeared in
Volume 95, Issue 24
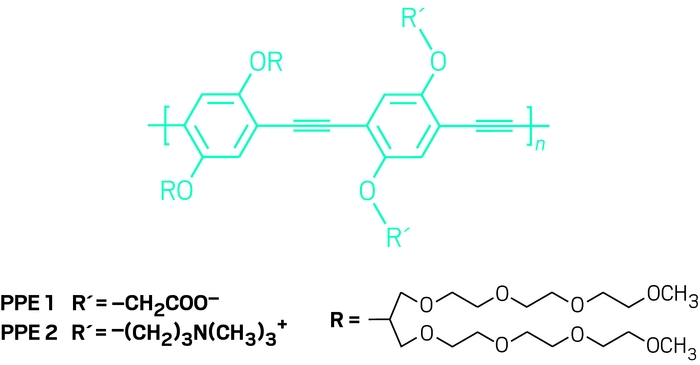
Whether you pay $50 or $5,000 for your whiskey, it’s safe to say most drinkers of the malted beverage don’t want to get duped into buying—let alone drinking—a counterfeit. An analytical technique that uses fluorescence modulation to discriminate between whiskeys could be a boon to the toolbox of techniques used by the beverage industry to catch fakes (Chem 2017, DOI: 10.1016/j.chempr.2017.04.008). The new strategy, which can distinguish whiskeys based on their brand, age, flavor, and region of origin, comes courtesy of Uwe H. F. Bunz of Heidelberg University and colleagues. The team previously developed a similar strategy for identifying white wine varieties. The researchers prepared “artificial tongue” polymer arrays based on 22 fluorescing charged and neutral poly(p-aryleneethynylene)s. They used these polymer arrays to assess 30 whiskeys, including single malts and blends, based on their signature fluorescence patterns created when interacting with the polymers. Three polymers stood out with the highest discriminative power: a positively charged molecule with a perfluorobenzylammonium group and two negatively charged molecules, one bearing carboxylic acid groups and the other equipped with sulfonate groups.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter