Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Microscopy
Cryo-electron microscopy innovators win 2017 Nobel Prize in Chemistry
Jacques Dubochet, Joachim Frank, and Richard Henderson honored for developing the technique, which enables unprecedented views of important biomolecules
by Stu Borman
October 4, 2017

The 2017 Nobel Prize in Chemistry has been awarded to Jacques Dubochet of the University of Lausanne, Joachim Frank of Columbia University, and Richard Henderson of the MRC Laboratory of Molecular Biology in Cambridge, England. They earned the prize “for developing cryo-electron microscopy for the high-resolution structure determination of biomolecules in solution.” The scientists will equally share the $1.11 million award.

In cryo-electron microscopy (cryo-EM), a beam of electrons is sent through a biomolecular sample that has been frozen, typically with liquid ethane. The material deflects the electrons in a way that permits researchers to determine the structure of the biomolecule. Electron beams physically damage biomolecules, but freezing them, the “cryo” part of cryo-EM, protects them from electron damage and prevents them from getting dehydrated in the electron microscope’s vacuum chamber.
Structures obtained by cryo-EM and related techniques are of fundamental importance for understanding life chemistry, and they can help scientists develop drugs by elucidating the way bioactive agents interact with biomolecules.
“Proteins are in all living things—humans, plants, animals, bacteria,” says Allison A. Campbell, president of ACS. The prize “highlights the role that chemistry plays in every aspect of our lives. From the medicines we take to the plants we grow, understanding proteins—and the chemistry of those proteins—is critical.”
Traditionally, scientists turned to X-ray crystallography and nuclear magnetic resonance spectroscopy to obtain biomolecular structures. Progress in cryo-EM, however, eventually enabled the technique to handle some structures that crystallography and NMR cannot. For example, it eliminates the need to crystallize biomolecules, which is required in crystallography and can be extremely difficult to accomplish in some cases.
Cryo-EM can also visualize larger structures than NMR or crystallography can: Cryo-EM specialist Sarah Butcher of the University of Helsinki estimates that the technique can analyze 100-fold larger structures, including whole viruses and even frozen cells. That makes it “much, much more adaptable to lots of different types of biological questions than NMR or X-ray crystallography,” she says.
Henderson set the groundwork for cryo-EM in 1975, when he used electron microscopy to determine a three-dimensional model of bacteriorhodopsin by averaging multiple images obtained with weak electron beams. That study showed that electron microscopy could obtain images as detailed as those determined by crystallography, which was an inherently higher-resolution technique at that time. During the next decade, Frank, then at the New York State Department of Health Wadsworth Center, developed image-processing technology for converting conventional 2-D electron microscopy pictures into 3-D structures. Henderson also helped advance image-processing techniques.
In the early 1980s Dubochet devised methods for rapidly freezing biomolecular samples to protect them from electron damage and dehydration while maintaining their native molecular shapes. In 1990, Henderson was able to obtain the first atomic-resolution cryo-EM structure—of bacteriorhodopsin, which has a well-ordered structure that made high resolution easier to achieve than would have been the case with many other biomolecules.
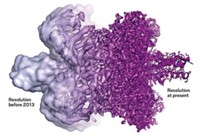
In the past decade, advances in electron-detection technology, particularly the development of direct electron detectors, have greatly improved cryo-EM’s resolution capabilities. The advent of these detectors, now commercially available, and improvements in data-handling techniques “caused a huge revolution in the quality of cryo-EM data,” Butcher says. “Now that means we can recover all the information and go to atomic resolution, which is what crystallographers do. But we can do it without having to make crystals, and crystals are a pain.”
Related stories:
Cryo-electron Microscopy Breaks The Crystal Ceiling
Crystallography Without Crystals
Uncovering the Spliceosome’s Secrets
Scientists solve Zika structure, identify possible fetal receptor
Cryo-electron tomography provides first view of a cell’s nucleus in its natural, undisturbed environment
New close-up views of the nuclear pore complex
NOTE: This story was updated on Oct. 5, 2017, to include new information.
This article has been translated into Spanish by Divulgame.org and can be found here.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter