Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Flipping from E- to Z-alkenes
Light-catalyzed reaction offers easy access to Z-isomer cross-coupling partners
by Tien Nguyen
March 5, 2018
| A version of this story appeared in
Volume 96, Issue 10

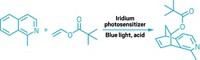
In their search for new medicines, chemists often choose the trusty Suzuki-Miyaura reaction to construct essential carbon-carbon bonds. This reliable reaction connects alkyl halides with various classes of organoborane compounds, including alkenyl organoboranes, which can be further classified by alkene configuration, either cis (Z) or trans (E). E-alkenylboron compounds are faster and simpler to make than their thermodynamically unfavored twin, the Z-configured compounds, and thus have received more attention as precursors for the Suzuki-Miyaura reaction. Now, using photoredox catalysis, researchers led by University of Münster’s Ryan Gilmour and University of St. Andrews’ Allan Watson are offering Z-alkenes a turn in the spotlight (Angew. Chem. Int. Ed. 2018, DOI: 10.1002/anie.201800286). Starting with the more accessible E-styrenyl organoboranes, the team employs an iridium catalyst and visible light to generate an excited state intermediate that may be swayed to collapse into either alkene conformation. An intentionally situated methyl substituent in the compound exerts the strain necessary to convince the molecule’s boron pinacol group to flip to the Z configuration. Though the reaction is currently limited to styrenyl boranes, Gilmour says it’s a good first step to providing access to Z-alkenyl organoboranes, which represent an underexplored area of chemical space for Suzuki-Miyaura cross-couplings.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter