Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Vaccines against addictive drugs push forward despite past failures
Vaccines could help prevent relapses but have yet to be effective in clinical trials
by Michael Torrice
February 19, 2018
| A version of this story appeared in
Volume 96, Issue 8
When Caron Block meets people who don’t understand the struggle of drug addiction, she asks them to imagine that someone has wrapped their head with saran wrap. “You are desperate for a breath; you’d do anything to get that saran wrap off your face so you can breathe,” Block says. That’s how a person dealing with substance abuse feels when having a craving, she explains. “So how horrible is it to have a craving like that and know at the same time that if you shoot up you could die?”
Block heard the saran wrap analogy while participating in a program at the Betty Ford Center that was designed for family members of those grappling with a substance use disorder.At the time, her son was receiving treatment for heroin addiction. He’s now 29 and has been sober for six years.
Once he got sober, Block started to look into possible medications or other treatments that could help prevent him from relapsing. “Because if AA were enough, or religion were enough, or ‘Just Say No’ were enough, people wouldn’t relapse and die,” she says, noting that heroin users who relapse have an increased risk of overdosing. “So I started doing my homework, and I found Kim Janda.”
Janda and his group at Scripps Research Institute California are working on an antiheroin vaccine. In practical terms that means a vaccine that would stimulate a person’s immune system to produce antibodies that bind to heroin. The antibodies would block the drug from crossing the bloodstream into the brain, stopping the person from experiencing a high and preventing a relapse.
The Scripps group is one of several working on vaccines against drugs of abuse, including cocaine and nicotine. These vaccines received a burst of media attention last year when Tom Price, then secretary of the U.S. Department of Health & Human Services, touted so-called vaccines for addiction as an “exciting prospect” in combating the current opioid epidemic.
Drug vaccines 101
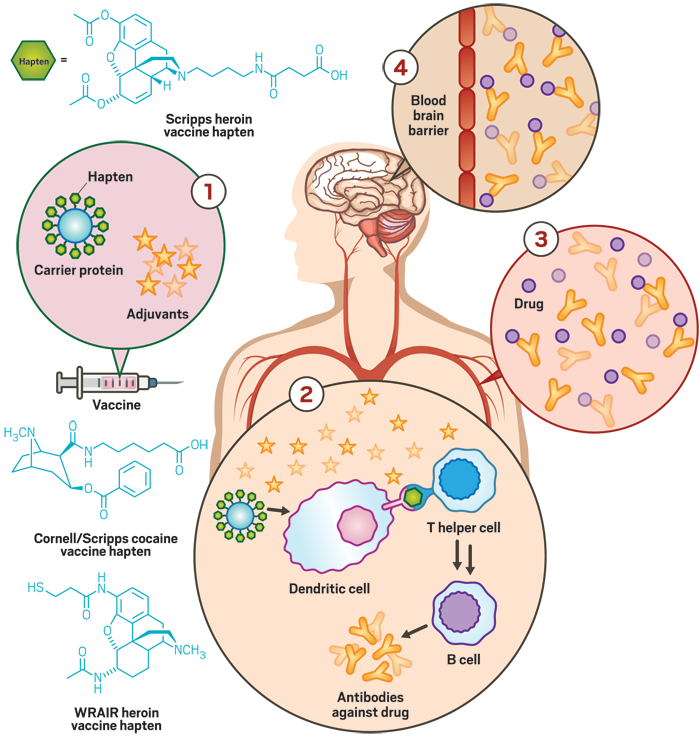
1. Vaccines against drugs of abuse consist of a carrier protein (blue) decorated with haptens (green), which are molecules that resemble the structures of the targeted drug. Adjuvants (yellow stars) help boost the immune system’s response to the vaccine.
2. In the bloodstream, immune cells called dendritic cells engulf the carrier protein conjugates, chew up the proteins, and display chunks, including the haptens on their surfaces. In lymph nodes, T helper cells get activated when they interact with the displayed chunks and then stimulate B cells to produce antibodies that bind to the drug. Adjuvants enhance the process by turning on pathways within the immune cells.
3. When a vaccinated person takes the drug, the generated antibodies bind to the drug molecules in the bloodstream.
4. The antibody-bound drug molecules can’t slip past the blood-brain barrier, so the drug can’t activate its target receptors in the brain.
But the idea of vaccinating people against drugs of abuse is decades old. Researchers have repeatedly validated the concept in animals but have yet to demonstrate a vaccine that is effective in people. Learning from the failures of past vaccines, scientists in the field have now developed new formulations that are closing in on or have already begun clinical trials.
Although interest from the pharmaceutical industry has been intermittent, experts in substance abuse treatment are guardedly hopeful. “The vaccines seem very promising, and they’re novel, providing a different mechanism to prevent substance abuse,” says Kelly E. Dunn, who studies opioid use disorders at Johns Hopkins University School of Medicine. “But there is still a lot of work to do.”
Breaking the cycle
For doctors, drug counselors, and therapists on the front lines of the opioid epidemic, new treatment options are greatly welcome, says R. Corey Waller, a practicing addiction specialist and chair of the legislative advocacy committee of the American Society of Addiction Medicine. “We’re looking for everything and anything,” he says. “We don’t care if it’s voodoo, unicorns, or rainbows; we’ll take it.”
Currently, few medical treatment options exist. For people addicted to heroin, morphine, and other opioids, methadone and buprenorphine can ease withdrawal symptoms and decrease cravings. These molecules activate the same brain receptor—the mu opioid receptor—as heroin and morphine. Rather than activate the receptor, another drug called naltrexone blocks the receptor and can decrease cravings over time.
But people struggling with addiction to cocaine or stimulants such as methamphetamine don’t have treatment options like these. They don’t exist. So Waller and Johns Hopkins’s Dunn think vaccines are particularly promising in treating people addicted to cocaine and stimulants.
The scientists working on drug vaccines see their work as offering a complementary approach to drug counseling and therapy, as well as to existing medical treatments. The vaccines may also offer advantages over existing medications in terms of cost and length of effect.
The main goal of the vaccines is to help people break the cycle of addiction. “Addicts will relapse,” Scripps’s Janda says. “What they want is just to have a little taste and then they spiral out of control and go on runs and they’re back to square one again.”
The crux of addiction is that drugs hijack the brain’s reward centers, Waller says. People no longer get rewarding feelings from normal activities, such as doing well at their jobs or hanging out with friends. Instead, they feel that sensation only when they take the drug.
The drug also establishes a reinforcing loop of behaviors triggered by sensory cues, Waller says. The cues can be seemingly small: Maybe it’s the sign on the person’s liquor store of choice or the type of car the dealer drives or the smell of the room that person used to shoot up in. These cues spark cravings, and when a person uses, the drug’s rewarding sensation strengthens the connection between the cue and the craving.
But this connection weakens over time if the person stops using. “The way the vaccine would work is if the person did use the drug, they wouldn’t get that reward, so it would decrease the body’s response to that cue the next time,” Waller says.
In the works
A partial list of vaccines against drugs of abuse currently in development.
| DRUG | DESCRIPTION | DEVELOPER | STATUS | REFERENCE |
|---|---|---|---|---|
| Cocaine | Cocaine haptens tethered to adenovirus capsid protein | Weill Cornell Medicine/Scripps Research Institute | Phase I | CNS Neurol. Disord.: Drug Targets 2011, DOI:10.2174/187152711799219334 |
| Nicotine | Nicotine haptens tethered to polymer nanoparticles encapsulating immune-stimulating molecules | Selecta Biosciences | Phase I | |
| Fentanyl | Fentanyl haptens tethered to tetanus toxoid; entolimod adjuvant | Baylor College of Medicine | Preclinical | |
| Fentanyl analogs | Fentanyl haptens tethered to tetanus toxoid | Scripps Research Institute | Preclinical | Angew. Chem. Int. Ed. 2016, DOI: 10.1002/anie.201511654 |
| Heroin | Heroin haptens tethered to tetanus toxoid | Scripps Research Institute | Preclinical | J. Am. Chem. Soc. 2017, DOI: 10.1021/jacs.7b03334 |
| Heroin | Heroin haptens tethered to tetanus toxoid; Army liposome formulation adjuvant | Walter Reed Army Institute of Research | Preclinical | J. Med. Chem. 2017, DOI: 10.1021/acs.jmedchem.7b01427 |
| Oxycodone | Oxycodone haptens tethered to keyhole limpet hemocyanin | Minneapolis Medical Research Foundation | Preclinical | PLOS One 2014, DOI: 10.1371/journal.pone.0096547 |
Making something small look big
The idea of preventing relapse by vaccinating people against drugs of abuse has been around since the 1970s. And the basic logic of the vaccines hasn’t changed much since then.
Normally, the immune system ignores small-molecule drugs in the bloodstream. The molecules are tiny enough that they sneak past the immune cells circulating in the body. To alert these cells to the presence of drug molecules, scientists must attach the compounds to larger things that already provoke the immune system.
Those larger things are called carrier proteins. The tetanus toxoid, which has a molecular weight of over 100 kilodaltons, is one common carrier protein, but other vaccines have used a cholera toxin or bovine serum albumin. A cocaine vaccine developed by Janda and Ronald G. Crystal’s group at Weill Cornell Medicine uses capsid proteins from adenovirus. And Selecta Biosciences has a nicotine vaccine that doesn’t use a protein, opting instead for polymer nanoparticles encapsulating immune-stimulating molecules.
Scientists decorate their carrier of choice with haptens—analogs of a drug such as heroin or cocaine that the immune system will develop antibodies against. Chemists design the hapten so that it contains key chemical features of the drug and then attach it to the carrier protein through a linker. To optimize the vaccine design, researchers evaluate a range of haptens that have the linker group attached to different positions on the molecule or that involve different conjugation chemistries. The key is to find the hapten and conjugation chemistry that yields a highly specific immune response against the drug when tested in an animal.
The third ingredient added to a vaccine against drug abuse is an adjuvant. Sometimes included in vaccines against pathogens such as hepatitis B, these compounds activate immune signaling pathways to rev up the cells involved in responding to the vaccine.
Once a person receives a vaccine, immune cells called dendritic cells engulf the hapten-protein conjugates, digest them, and display chunks of them, including the hapten, on their surfaces. Other immune cells start to interact with these displayed chunks and trigger the production of antibodies that can bind drug molecules that look like the hapten.
Vaccines against pathogens such as the hepatitis B virus work by establishing immune cells that remember the molecular details of a threat. If the pathogen arrives in the body, these cells recognize it and mount a response to it.
But vaccines against drugs of abuse must work differently because once the drug is in the bloodstream, it can immediately enter the brain and activate its target receptors to produce a high. So high levels of antibodies are key to the success of a drug vaccine. “With an infectious disease, you usually have time for the immune system to respond before the infection takes root,” says Gary R. Matyas, who works on a heroin vaccine at Walter Reed Army Institute of Research (WRAIR). “But in the case of a drug of abuse, you need to be ready for the drug at the time of injection. You need high levels of the antibody and keep them high.”
Some skeptics of the approach point out that people who want to get high could just take more drug to overwhelm the antibodies elicited by a vaccine. Vaccine developers don’t deny that is possible, but they argue that these treatments are designed for people dedicated to quitting and looking for help to prevent relapse.
So far, scientists have repeatedly demonstrated that the vaccine concept works in animals. These vaccines establish high antibody levels for months in animals, and these antibodies can block a drug’s action. For example, when injected with heroin, vaccinated rodents don’t experience the drug’s pain-relieving properties. One imaging study showed that cocaine failed to bind to its molecular target in the brains of monkeys vaccinated against cocaine (Neuropsychopharmacology 2013, DOI: 10.1038/npp.2013.114). And in multiple studies, the vaccines have stopped animals from self-administering drugs.
“If you are an experimental animal, we have very effective vaccines for you,” Crystal says. “But humans aren’t just big mice or big monkeys.”
Learning from failure
About a decade ago, a handful of vaccines—against nicotine and cocaine—entered clinical trials. “All of them were disappointing,” says Ivan Montoya of the National Institute on Drug Abuse.
None of the trials demonstrated a significant reduction in drug use for people receiving the vaccines compared with those getting placebo shots. Within the trials’ data, however, lies a lesson for the field.
Not all the participants receiving the vaccine reacted similarly. Some people’s immune systems produced high levels of antibodies against the drug, and others did not. Data for people with the highest antibody levels suggested that the vaccine had decreased drug use. “We learned we needed to make more antibodies in a greater percentage of people,” says Thomas R. Kosten, who develops vaccines at Baylor College of Medicine.
On the basis of that conclusion, researchers started optimizing all three components of their vaccines to elicit higher antibody levels. Some are trying new immune-stimulating carriers and adjuvants, while others have sought more chemically stable haptens and conjugation chemistries.
WRAIR’s Matyas and his team are betting on a more potent adjuvant called Army liposome formulation (ALF) to help their heroin vaccine. ALF consists of spherical sacs of lipids—liposomes—that include monophosphoryl lipid A, a component of Gram-negative bacterial walls. When immune cells engulf the liposomes, monophosphoryl lipid A activates numerous immune pathways, including those that involve toll-like receptor 4. Meanwhile, Kosten is working with a protein-based adjuvant called entolimod for his group’s vaccine against fentanyl, a powerful opioid. Entolimod is a modified version of a Salmonella flagellin protein that turns on another toll-like receptor.
Others in the field are seeking biomarkers to identify people who may not respond sufficiently to a vaccine. “We learned that about 10% of people are not going to ever generate sufficient antibodies,” Kosten says.
Researchers will have to wait for clinical trial data to find out if these new formulations work. Selecta has started a Phase I clinical trial of its nicotine vaccine, and Weill Cornell’s Crystal started one for the adenovirus-based cocaine vaccine. Four groups have opioid vaccines that haven’t yet made it to the clinic. Janda and WRAIR have heroin vaccines, Marco Pravetoni and Paul R. Pentel of the Minneapolis Medical Research Foundation have an oxycodone vaccine, Kosten has one against fentanyl, and Janda also has one against fentanyl analogs.
Advertisement
While National Institute on Drug Abuse funding has helped most of these groups formulate new vaccines in the research lab, some of the opioid vaccine groups are struggling to secure enough money to do the appropriate safety testing and vaccine production required to meet U.S. Food & Drug Administration standards to start clinical trials. For that kind of funding, these groups have turned to industry with limited success.
Bernard Munos, a senior fellow at FasterCures, a think tank at the Milken Institute focused on accelerating and improving medical research, says pharmaceutical companies are hesitant to fund research into addiction therapies such as these vaccines because they don’t see a viable market. “Even if an effective drug or vaccine were approved tomorrow, who would be the customer?” he asks. Patients exist, he says, but the logistics of treating people with substance use disorders are complicated enough to scare away companies.
To create a market, Munos thinks the government will have to get involved. Cash prizes and advance purchase commitments for therapies that demonstrate effectiveness could help prime the pump to get companies interested, he says. “Given the enormous burden of drug addiction to society, the lack of a commercial market should not be a showstopper,” Munos adds. “We must try new approaches.”
The opioid vaccine researchers think that once they do secure sufficient funding, getting their vaccines into clinical trials won’t be a problem.
Caron Block has been fundraising to help Janda’s heroin vaccine make it to clinical trials, and she hopes all the vaccines succeed. She sees them as a way for people struggling with sobriety to get a second chance. “Maybe they get a moment of clarity where they can say, ‘Boy I was lucky. I should get help.’ ”.
CORRECTION: The story was updated on Feb. 20, 2018, to correct the reference for the oxycodone vaccine entry in the table.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter