Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Neuroscience
Psychedelics forge connections between neurons
Study in rats and fruit flies suggests that these drugs could inspire better, ketamine-like antidepressants
by Cici Zhang
June 13, 2018
| A version of this story appeared in
Volume 96, Issue 25

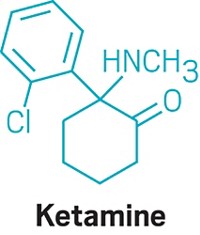
About one-third of people with depression do not find relief from their symptoms with current drugs for the disease. In recent years, psychiatrists have become interested in using low doses of the anesthetic ketamine as an antidepressant because it often works in patients who don’t respond to conventional drugs and it acts fast—having an effect within hours instead of weeks or even months. But the drug has undesirable side effects. It can produce out-of-body feelings and hallucinations, and it has the potential for abuse. Scientists would like to understand how ketamine works as an antidepressant so they can design new molecules that lack the negative aspects of the anesthetic.

Now, a study reports that other psychedelic compounds have similar effects on neurons as ketamine does: They promote the growth of connections between neurons (Cell Rep. 2018, DOI: 101016/j.celrep.2018.05.022). The work suggests new chemical scaffolds that could mimic ketamine’s antidepressant properties, says lead author David Olson of the University of California, Davis.
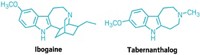
Previous research had shown that ketamine can rapidly grow synapses—the connections between neurons—in brain areas that regulate emotion and mood, possibly accounting for the drug’s fast-acting therapeutic effects. In the current study, the UC Davis team found that several psychedelic compounds—for example, N,N-dimethyltryptamine (DMT), 2,5-dimethoxy-4-iodoamphetamine (DOI), and lysergic acid diethylamide (LSD)—increased the number of synaptic connections in the brains of rats and fruit flies, as well as in cultured neurons from the animals. As with ketamine, the effects of the drugs were long lasting. When the researchers injected rats with DMT, which is found in an Amazonian herbal tea called ayahuasca, they still observed synaptic changes 24 hours later, well after the animals had cleared the drug from their bodies, Olson says. The team previously had demonstrated that DMT produced antidepressant-like effects and stopped behaviors that resemble posttraumatic stress disorder in rats (ACS Chem Neurosci. 2018, DOI: 10.1021/acschemneuro.8b00134).
In the new study, the scientists also determined that these other psychedelics promote synaptic growth through a similar signaling pathway involving the protein mTOR that ketamine does. Ronald Duman, a neuroscientist at Yale University who studies ketamine, says that the findings are “interesting and important” because they could explain how these agents might treat mood disorders. While ketamine activates the mTOR pathway by blocking N-methyl-D-aspartate receptors on neurons, the psychedelic compounds do so via activating a different target, the 5-HT2A receptor, which according to Duman suggests a novel approach for treating depression.
Roland Griffiths, a neuroscientist at Johns Hopkins University School of Medicine agrees that the new study is important. “Almost nothing is known about the neural mechanisms underlying the ability of psychedelics to produce enduring change in moods, attitudes, and behavior”he says.
But David Feifel, a professor emeritus of psychiatry at University of California, San Diego, who has used ketamine to treat patients with depression, says more work is needed before the reported mechanisms can be identified as those responsible for these drugs’ antidepressant properties. The next step, he says, would be to block the pathway outlined in this study and then see if that prevents the drugs from producing an antidepressant effect in animals.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter