Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthetic Biology
Artificial metalloenzyme flips switch in cells
Cell-penetrating assembly uncages hormone, turning on gene circuit
by Celia Arnaud
May 23, 2018
| A version of this story appeared in
Volume 96, Issue 22

By adding artificial metalloenzymes to cells, researchers can coax the cells to do a job they can’t normally do. Typically, that out-of-the-ordinary task is fairly simple, like uncaging a fluorescent reporter molecule.
Swiss researchers, led by Thomas R. Ward of the University of Basel; Martin Fussenegger of Swiss Federal Institute of Technology, Zurich; and Stefan Matile of the University of Geneva, decided to take artificial metalloenzymes to the next level. Their synthetic enzyme uncages a molecule that then switches on a synthetic gene circuit inside cells (Nat. Commun. 2018, DOI: 10.1038/s41467-018-04440-0). Such an approach might one day allow researchers to use designer cells to produce protein therapeutics.
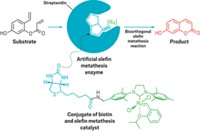
The new metalloenzyme comprises three main components: the protein streptavidin, a ruthenium catalyst, and a cell-penetrating polymer. Streptavidin serves as a scaffold to which the researchers attach the catalyst and the polymer via biotin chemistry. The catalyst helps cleave, or uncage, a molecule containing an allyl-carbamate protecting group, and the polymer helps the whole assembly slip into the cell.
The researchers added the metalloenzyme to human kidney cells engineered to contain a gene circuit that responds to thyroid hormone. They separately added thyroid hormone that was caged with an allyl-carbamate protecting group. No known natural enzymes cleave allyl carbamates, so the only way to free the hormone is with the artificial metalloenzyme. After the hormone is released, it activates the gene circuit, designed to produce the enzyme luciferase, which can trigger a light-emitting reaction. Seeing light emitted by the kidney cells confirmed for the researchers that the metalloenzyme had done its new job.
Getting artificial metalloenzymes to work inside cells is challenging “because many cellular components can inhibit the biocatalysts,” says Yi Lu of the University of Illinois, Urbana-Champaign, who also designs artificial metalloenzymes. The new work represents “a major step toward engineering interesting and potentially novel functions in living cells using highly modular cell-penetrating artificial metalloenzymes,” he says.
Scientists have previously uncaged molecules with synthetic metalloenzymes, says Paul A. Wender of Stanford University. “This work adds to that by using a cell-penetrating subunit to carry the catalyst into cells,” adds Wender, who has also worked on bioorthogonal catalysts and cell-penetrating systems. “The choreography exhibited in this cascade is stunning and bodes well for future advances in this emerging field. Congratulations to the authors for this impressive contribution.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter