Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Athina Anastasaki
This materials chemist shows prowess at pulling polymers apart
by Bethany Halford
May 19, 2023
| A version of this story appeared in
Volume 101, Issue 16

Credit: Anastasia Besika/Will Ludiwg/C&EN | Athina Anastasaki
thina Anastasaki shares her name with the Greek goddess of wisdom and war, so it might be tempting to ascribe her chemical acumen to some divine source. But she attributes her success in science to a lifelong love of learning and the guidance of good mentors rather than any intervention from mythical figures.
Anastasaki grew up in Athens, Greece, and spent her summers swimming and hiking on the island of Crete, where her parents were from. “I spent my best part of the year in Crete—I feel this is part of my soul,” she says. Anastasaki recalls a childhood spent looking over X-ray films with her mother, a radiologist, and doing schoolwork alongside her father, a geology professor, as he kept up with the literature in his field. This academic atmosphere meant that she learned to like studying—“but not because I was told to study,” she says.
A high school teacher sparked Anastasaki’s interest in chemistry. “I’m always inspired by people rather than subjects,” she says. She decided on a career as a high school chemistry teacher and studied chemistry and polymer science as an undergraduate at the National and Kapodistrian University of Athens. Thinking a doctoral degree and postdoctoral experience would give her an edge in the job market, she pursued a PhD with Dave Haddleton at the University of Warwick. Later, she completed postdoctoral studies—first with Tom Davis at Monash University and later with Craig Hawker at the University of California, Santa Barbara.
Advertisement
During her studies, Anastasaki discovered that she enjoys research as much as teaching. “My true love turns out to be polymer synthesis,” she says. In 2019, she returned to Europe to become a professor of polymeric materials at the Swiss Federal Institute of Technology (ETH), Zurich.
There, she has taken on the challenge of designing polymers that can be readily broken into their most basic building blocks so that at the end of their lives they can be recycled to make high-value materials. Typically, when polymers are recycled, they’re made into materials with lower-end applications because current recycling technologies degrade the polymer to some extent. Anastasaki’s strategy allows chemists to regenerate the starting monomer so that it can be reused to make polymers that are as good as new.
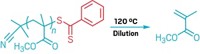
She recently demonstrated this method by creating polymethacrylates—the type of polymer that makes up plexiglass—and then pulling the polymer apart by simply heating it to 120 °C. The key was to use a type of radical-driven polymerization called reversible addition-fragmentation chain transfer (RAFT), which puts special chemical groups at the ends of the polymer chains. These end groups, such as dithiobenzoate, are primed to trigger the depolymerization process without needing a catalyst or extreme temperatures.
“My true love turns out to be polymer synthesis.”
—Athina Anastasaki, professor, Swiss Federal Institute of Technology (ETH), Zurich
Industry has been slow to adopt RAFT polymerization, in part because the end groups add to the costs of the polymerization process, Anastasaki says. But her group has been able to recover the end groups after depolymerization, a step she thinks will make the process less expensive and more attractive to companies. “I’m hoping that this could encourage further industrial implementation,” she says.
Anastasaki’s group is now working on controlled depolymerization, which would allow chemists to depolymerize a material by degrees rather than go all the way back to the monomer. This could save energy if a polymer maker wanted to take a high-molecular-weight polymer and turn it into a material with lower viscosity for a different application.
Anastasaki hopes to someday develop depolymerization methods that can be applied to some of the world’s most persistent polymers, like polystyrene. “Everyone says that it’s impossible to chemically recycle materials like polystyrene. I think it’s possible,” she says.
Plastics and polymers can last in the environment for a very long time, says Erick M. Carreira, a colleague of Anastasaki’s at ETH Zurich. “The idea that she’s tackling a problem involving how to think about degrading these things, I thought was a phenomenal choice,” he says. What’s more, Carreira says, her approach involves thinking “at a fundamental level about the chemistry that would be required to reverse the polymerization process.”
Tackling the global plastic waste challenge requires a mix of bold action and strategic planning, traits Anastasaki shares with her eponymous goddess. As Anastasaki says: “There’s some benefit in combining the spirit of fighting with wisdom.”
Vitals
Current affiliation:
Swiss Federal Institute of Technology (ETH), Zurich
Age: 35
PhD alma mater:
University of Warwick
Hometown:
Athens, Greece
My lab superpower is:
“I am better at designing experiments than executing them. My students are my lab superpower, as I don’t need to show them how to work at the bench!"
My favorite book is:
“The Count of Monte Cristo. I read it for the first time when I was 8 years old. I very much admire the hero, as he manages to retain his faith throughout a series of adversities and finds the strength to forgive himself and others and seeks redemption and happiness.”
Learn more/nominate a rising early-career chemist to be one of C&EN's Talented 12 at:
cenm.ag/t12-nominations-2024
Athina Anastasaki shares her name with the Greek goddess of wisdom and war, so it might be tempting to ascribe her chemical acumen to some divine source. But she attributes her success in science to a lifelong love of learning and the guidance of good mentors rather than any intervention from mythical figures.
Vitals
▸ Current affiliation: Swiss Federal Institute of Technology (ETH), Zurich
▸ Age: 35
▸ PhD alma mater: University of Warwick
▸ Hometown: Athens, Greece
▸ My lab superpower is: “I am better at designing experiments than executing them. My students are my lab superpower, as I don’t need to show them how to work at the bench!”
▸ My favorite book is: “The Count of Monte Cristo. I read it for the first time when I was 8 years old. I very much admire the hero, as he manages to retain his faith throughout a series of adversities and finds the strength to forgive himself and others and seeks redemption and happiness.”
Anastasaki grew up in Athens, Greece, and spent her summers swimming and hiking on the island of Crete, where her parents were from. “I spent my best part of the year in Crete—I feel this is part of my soul,” she says. Anastasaki recalls a childhood spent looking over X-ray films with her mother, a radiologist, and doing schoolwork alongside her father, a geology professor, as he kept up with the literature in his field. This academic atmosphere meant that she learned to like studying—“but not because I was told to study,” she says.
A high school teacher sparked Anastasaki’s interest in chemistry. “I’m always inspired by people rather than subjects,” she says. She decided on a career as a high school chemistry teacher and studied chemistry and polymer science as an undergraduate at the National and Kapodistrian University of Athens. Thinking a doctoral degree and postdoctoral experience would give her an edge in the job market, she pursued a PhD with Dave Haddleton at the University of Warwick. Later, she completed postdoctoral studies—first with Tom Davis at Monash University and later with Craig Hawker at the University of California, Santa Barbara.
During her studies, Anastasaki discovered that she enjoys research as much as teaching. “My true love turns out to be polymer synthesis,” she says. In 2019, she returned to Europe to become a professor of polymeric materials at the Swiss Federal Institute of Technology (ETH), Zurich.
There, she has taken on the challenge of designing polymers that can be readily broken into their most basic building blocks so that at the end of their lives they can be recycled to make high-value materials. Typically, when polymers are recycled, they’re made into materials with lower-end applications because current recycling technologies degrade the polymer to some extent. Anastasaki’s strategy allows chemists to regenerate the starting monomer so that it can be reused to make polymers that are as good as new.
She recently demonstrated this method by creating polymethacrylates—the type of polymer that makes up plexiglass—and then pulling the polymer apart by simply heating it to 120 °C. The key was to use a type of radical-driven polymerization called reversible addition-fragmentation chain transfer (RAFT), which puts special chemical groups at the ends of the polymer chains. These end groups, such as dithiobenzoate, are primed to trigger the depolymerization process without needing a catalyst or extreme temperatures.
Industry has been slow to adopt RAFT polymerization, in part because the end groups add to the costs of the polymerization process, Anastasaki says. But her group has been able to recover the end groups after depolymerization, a step she thinks will make the process less expensive and more attractive to companies. “I’m hoping that this could encourage further industrial implementation,” she says.
Anastasaki’s group is now working on controlled depolymerization, which would allow chemists to depolymerize a material by degrees rather than go all the way back to the monomer. This could save energy if a polymer maker wanted to take a high-molecular-weight polymer and turn it into a material with lower viscosity for a different application.
Anastasaki hopes to someday develop depolymerization methods that can be applied to some of the world’s most persistent polymers, like polystyrene. “Everyone says that it’s impossible to chemically recycle materials like polystyrene. I think it’s possible,” she says.
Plastics and polymers can last in the environment for a very long time, says Erick M. Carreira, a colleague of Anastasaki’s at ETH Zurich. “The idea that she’s tackling a problem involving how to think about degrading these things, I thought was a phenomenal choice,” he says. What’s more, Carreira says, her approach involves thinking “at a fundamental level about the chemistry that would be required to reverse the polymerization process.”
Tackling the global plastic waste challenge requires a mix of bold action and strategic planning, traits Anastasaki shares with her eponymous goddess. As Anastasaki says: “There’s some benefit in combining the spirit of fighting with wisdom.”



















Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter