Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Energy Storage
Degradable battery ditches the metals
Polypeptide electrodes decompose to amino acids
by Leigh Krietsch Boerner
May 6, 2021
| A version of this story appeared in
Volume 99, Issue 17

The lithium-ion batteries that power small electronics are rarely recycled. These materials also use significant amounts of cobalt, which is in short supply and sometimes mined using unethical practices. Jodie Lutkenhaus, Karen Wooley, and coworkers at Texas A&M University have now built a battery that avoids these problems. Their metal-free battery uses an organic polypeptide backbone for its electrodes, which the scientists can degrade with acid into amino acids for potential reuse (Nature 2021, DOI: 10.1038/s41586-021-03399-1).
The design is a step toward a circular battery life cycle, in which batteries could be degraded and rebuilt without entering the waste stream. This offers an improvement over batteries made from nondegradable organic polymers, another approach for eliminating metals from batteries. These organic polymer batteries would likely have the same fate as other plastics, Wooley says. “More than 90% of the plastics that could be recycled are not.”
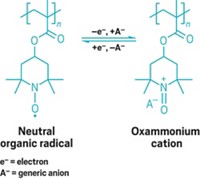
To make a battery that could reliably store charge without metals, the scientists had to completely rethink the chemistry, Lutkenhaus says. The team started with
To test the battery’s degradation, the team added hydrochloric acid and used mass spectrometry to watch the compound fall apart into
The authors’ design of a polypeptide organic battery that can degrade to its raw materials fills a need for biodegradable pathways, chemistry that’s lacking in current technologies, says Natia Frank, an organic materials chemist at the University of Nevada, Reno.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter