Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Nanomaterials
TiO₂ nanocrystals exhibit unusual slow-motion blinking
Made via template-based synthesis, the defective crystals actively mediate photocatalysis
by Mitch Jacoby
June 13, 2020
| A version of this story appeared in
Volume 98, Issue 23
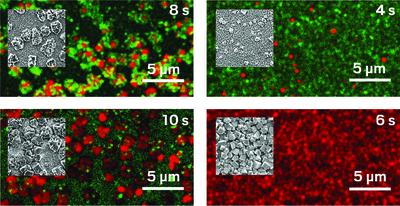
Like microscopic fireflies, semiconductor nanocrystals, also known as quantum dots, light up intermittently. For more than 20 years, researchers have worked to understand and control these random fluctuations in light emission because the “blinking” limits the stability of quantum dot–based devices like solar cells and light-emitting diodes. The observation of what may be a new type of blinking behavior suggests that researchers have more work to do (Angew. Chem., Int. Ed. 2020, DOI: 10.1002/anie.202005143). A team led by Tao Zhang, Tewodros Asefa, and Alexei M. Tyryshkin of Rutgers University and Eliška Mikmeková of the Czech Academy of Sciences reports that treating a polymer-derived porous carbon template with a titanium dioxide precursor yields TiO2 nanocrystals—limited to 10 nm in diameter and riddled with oxygen vacancies—that blink in an unprecedented way. In contrast to years of studies reporting rapid light–induced blinking with flashes often lasting just a fraction of a second—sometimes longer—the new work describes electron beam–induced flashes lasting roughly 15 s. In addition, when the crystals are irradiated with light, the resulting charge separation is unusually stable, making the particles active photocatalysts, as shown by tests in which the team used them to reduce carbon dioxide with water.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter