Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Antibiotics
Conjugate sneaks antibiotic into Gram-negative bacteria
Molecular Trojan horse tricks microbes into unleashing weapon of their own destruction
by Celia Henry Arnaud
April 4, 2018
| A version of this story appeared in
Volume 96, Issue 15

A new drug conjugate tricks Gram-negative bacteria into succumbing to an antibiotic that usually works only against Gram-positive bacteria.
Gram-negative bacterial infections are tough to treat because the microbes have an extra outer membrane that is hard for antibiotics to traverse. And the ones that do get in are usually pumped right back out by the cells.
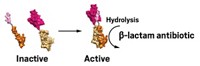
To kill these hardy bacteria, Marvin J. Miller and coworkers at the University of Notre Dame have put a twist on a type of drug conjugate called a sideromycin. Such combinations usually consist of a siderophore—a chelating agent bacteria use to grab and collect iron from their surroundings—connected to an antibiotic. Usually there’s only one antibiotic attached, but Miller’s team adds a second one. Their synthetic sideromycin includes a siderophore, a cephalosporin, and an oxazolidinone (J. Med. Chem. 2018, DOI: 10.1021/acs.jmedchem.8b00218).
The siderophore gets a bacterium to take up the conjugate. Then the microbe degrades the cephalosporin linker with a β-lactamase enzyme, releasing the oxazolidinone. That enzyme is usually part of the bacterium’s defense against antibiotics, but in this case, it unleashes the weapon of its own destruction.
Oxazolidinones usually work only against Gram-positive bacteria. The Notre Dame team shows that once the antibiotics breach the microbes’ defenses, they can also kill Gram-negative bacteria, including Acinetobacter baumannii, Escherichia coli, and Pseudomonas aeruginosa.
The work “highlights the remarkable ability of siderophore transport machinery to deliver complex molecules and provides a terrific example of how microbial enzymes associated with virulence and antibiotic resistance can be leveraged for new therapeutic strategies,” says Elizabeth M. Nolan of MIT, who also studies siderophore-antibiotic conjugates but wasn’t involved with the new work. “It will be important to determine whether this molecular design provides antibacterial activity against other Gram-negative bacterial pathogens that express β-lactamases.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter