Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Nanomedicines
Nanoparticle mouthwash could prevent tooth decay
Iron oxide particles and hydrogen peroxide bust biofilms on teeth in rat study
by Katharine Sanderson, special to C&EN
August 1, 2018
| A version of this story appeared in
Volume 96, Issue 32
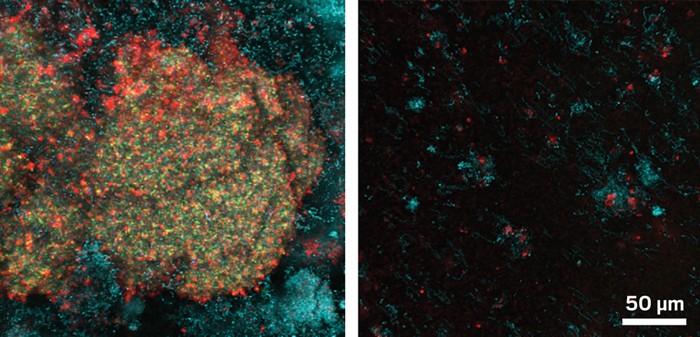
Sloshing a mouthwash of iron oxide nanoparticles around your mouth could someday save you a trip to the dentist. A new study in rats demonstrates that such a mouthwash can prevent and stop cavities by disrupting destructive bacterial biofilms that grow on teeth (Nat. Comm. 2018, DOI: 10.1038/s41467-018-05342).
The researchers, led by Hyun (Michel) Koo from the University of Pennsylvania School of Dental Medicine, used a formulation known as ferumoxytol, which consists of iron oxide nanoparticles coated with carboxymethyl dextran. Fermuoxytol is approved by the U.S. Food & Drug Administration for treating iron deficiency.
The team decided to test the teeth-protecting abilities of these iron oxide nanoparticles because they can break down hydrogen peroxide to create hydroxyl radicals that can disrupt biofilms. Koo and colleagues devised a test mouthwash therapy involving solutions of ferumoxytol and hydrogen peroxide. They used nanoparticle doses that were less than 1% of what is used to treat iron-deficiency, Koo says.
Twice a day over a period of three weeks, the researchers used both solutions to rinse the mouths of rats with cavities that mimic extreme childhood dental decay. Compared with animals that didn’t get the mouthwash, treated rats saw significant improvements in their dental decay. The mouthwash both stopped the progression of existing damage and prevented damage to smooth, healthy teeth.
The approach shows promise, says Zainab Hosseini-Doust, a bioengineer at McMaster University. “In most cases, the only effective way of eradicating a biofilm is physical removal of the infected biomaterial,” she says. “What’s interesting about this work is the ability of the developed nanoparticles to degrade the biofilm matrix from within.”
Hosseini-Doust would like to know more about how the nanoparticles interact with healthy oral biofilms. “The health of our teeth and oral cavity is highly dependent on the community of good bacteria living in our mouth. Any nonspecific antibacterial treatment will inevitably affect our good bacteria,” she says.
The team is now working to develop formulations with higher catalytic activity and, eventually, to test the efficacy of the mouthwash in humans, Koo says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter