Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Catalysis
Catalyst deconstructs polyethylene, producing valuable alkanes
Porous material mimics enzymes that repeatedly snip segments off proteins and other long-chain biomolecules
by Mitch Jacoby
October 14, 2020
| A version of this story appeared in
Volume 98, Issue 40
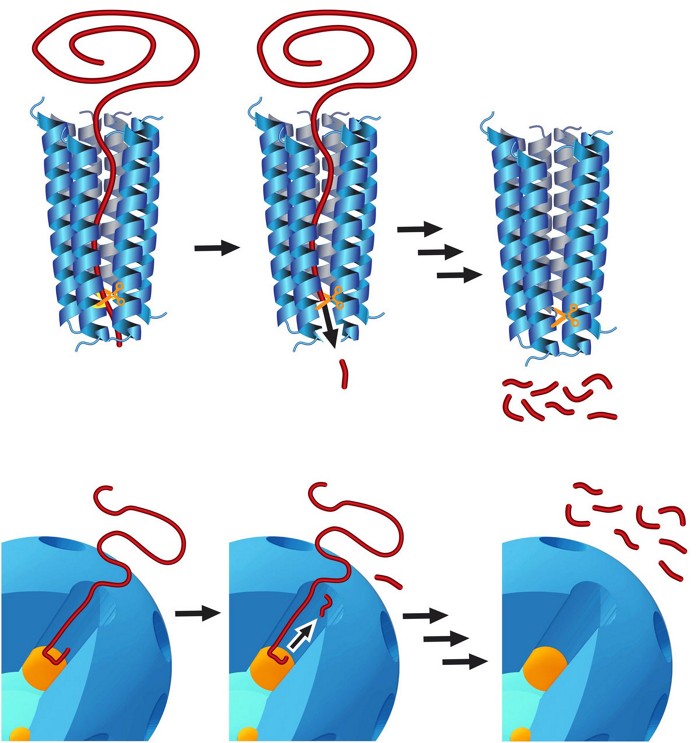
A catalytic system consisting of porous silica and platinum nanoparticles can convert high-density polyethylene to compounds used for making diesel fuel and lubricants (Nat. Catal. 2020, DOI: 10.1038/s41929-020-00519-4).
The synthetic system mimics natural enzymes that digest proteins by repeatedly snipping the molecular chains. It could provide a way to turn plastic waste into products that are more valuable than those made via melt-processing and other methods for recycling plastics.
To make the catalyst, researchers led by Wenyu Huang, Aaron D. Sadow, and Frédéric A. Perras of Ames Laboratory deposited roughly 3 nm diameter platinum nanoparticles on larger amine-functionalized silica spheres. Then they grew a shell of silica riddled with nanometer-sized pores around the silica spheres.
NMR spectroscopy and other types of analyses show that the structure of the catalyst, the length and width of its pores, and polymer-surface interactions drive polyethylene chains to snake their way into the narrow pores. At the bottom of the pores, the chains encounter the catalytic Pt nanoparticles and undergo hydrogenolysis, which snips off a well-defined segment. The segment exits the pore as the chain continues to crawl in, successively being chopped and shortened.
Catalysis tests using various types of plastics, including those used to make grocery-style shopping bags, show that the catalyst and breakdown procedure can be tuned to produce a narrow range of alkanes commonly used in diesel fuel and lubricants.
Gregg T. Beckham of the National Renewable Energy Laboratory describes the study as “an elegant combination of catalyst design, spectroscopy, and polymer science.” He expects the work will lead to new catalyst designs that use porous materials for polymer deconstruction.
The University of Minnesota’s Marc A. Hillmyer, a polymer specialist, says the tailored-pore approach is “quite innovative,” but follow-up work needs to be done. “Moving from demonstration systems to practical waste streams will be a valuable next step,” he adds.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter