Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Catalysis
Electrochemical method converts CO2 to graphite-like solids and other forms of carbon
Liquid-metal-based catalyst containing cerium functions at room temperature and resists deactivation
by Mitch Jacoby
March 3, 2019
| A version of this story appeared in
Volume 97, Issue 9
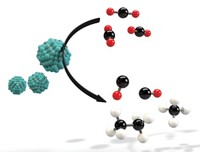
Driven to reduce atmospheric levels of greenhouse gases, researchers worldwide are developing methods for sequestering CO2, including injecting the gas underground and converting it to valuable liquid fuels and chemicals. But subterranean storage is expensive, and converted carbon could end up back in the atmosphere if it’s turned into volatile liquids or fuels that will release CO2 anew when burned. So Dorna Esrafilzadeh and Torben Daeneke of the University of New South Wales and coworkers developed a low-cost electrocatalytic method for converting CO2 to solid carbon, a nonvolatile material that can be used to make electrodes for energy storage or be safely buried (Nat. Commun. 2019, DOI: 10.1038/s41467-019-08824-8). To perform the reaction, the team fashioned an electrochemical cell featuring an unusual electrode. Its tip is made of a liquid-metal Ga-In-Sn alloy dosed with elemental cerium nanoparticles that make it catalytically active. Unlike a solid Ga-Ce catalyst, which quickly deactivates as a carbon layer fouls its surface, the team’s liquid catalyst resists fouling and remains stable. Using the test cell at room temperature and low voltage, the team converted CO2 to porous, graphite-like solids and showed that the materials work well as electrodes for high-efficiency capacitors.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter