Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Medicinal Chemistry
Photoswitchable drugs could light the way to more targeted treatments
Compounds switched on and off with light may limit side effects when used against cancer, blindness, and other disorders
by Katharine Sanderson
April 2, 2018
| A version of this story appeared in
Volume 96, Issue 14
In the fight against cancer, our therapeutic options have improved over the years. But they’re still far from perfect. Today, doctors administer chemotherapy drugs in hopes of killing a tumor before doing too much damage to other parts of a person’s body.
Now, chemists are hoping to gain even more ground in the battle, by designing light-activated small molecules that can be switched on in the body only where they’re needed—in the tumor—and off everywhere else, to limit side effects.
Not only are scientists aiming to use these photoswitchable drugs to fight cancer, but they also think the designer compounds could be useful in treating blindness and other disorders. Photoswitchable antibiotics might even help us fight drug resistance. By keeping the compounds deactivated when they’re not hard at work in the body fighting infection, we could prevent pathogens from building tolerance if the molecules spread into the environment.
Although interest in developing these photoswitchable compounds is on the rise, the field still has significant research issues to address. For instance, scientists need to ensure that molecular switches work at clinically useful wavelengths—those that can pass through tissue without doing damage. These scientists also need to work closely with clinicians to find practical ways of coupling light into a person’s body to reach various target zones.
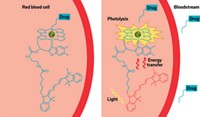
The concept of using light to treat disease isn’t completely new. For example, photodynamic therapy (PDT) treats skin cancers and other skin conditions. In PDT, people receive dye molecules as pills, injections, or creams applied to the skin. Doctors shine light onto the affected skin to activate the dye molecules, which then convert oxygen in the body’s tissue into toxic singlet oxygen that kills off diseased cells.
But PDT’s reliance on the body’s oxygen has its drawbacks: Tumors are often hypoxic—starved of oxygen—so the strategy can have diminished potency.

Photoswitchable therapies, part of a field often dubbed photopharmacology, have a different fundamental mechanism that might overcome this limitation. Unlike PDT dye molecules, photoswitchable therapies “take the energy from a photon to drive a chemical change within their own structure,” explains Edith C. (Phoebe) Glazer at the University of Kentucky. And that new structure binds to a particular target in cells to cause toxicity.
Glazer develops so-called photoactivated chemotherapy (PACT) drugs that function as PDT sensitizers but also as one-way photoswitches. When activated with light, her research team’s ruthenium(II) polypyridyl complexes irreversibly eject a methylated ligand. This ejection enables the complexes to cross-link DNA, inflicting cell damage wherever they’ve been illuminated. By modifying the PACT drugs’ ligands, Glazer can tune the molecules’ solubility and the wavelengths of light that they absorb, or make “dual action” compounds that both eject ligands and generate singlet oxygen (J. Am. Chem. Soc. 2012, DOI: 10.1021/ja3009677).
These molecules can be really potent, Glazer says. “Cells have evolved to deal with singlet oxygen, since this occurs naturally, but metal-DNA adducts do not occur naturally, so this can be more challenging for the cell to repair,” she explains.
But many chemists, like Wiktor Szymanski at University Medical Center Groningen, want to go one step further and use light to switch drugs on and then off again. His focus recently has been on photoswitchable antibiotics. Szymanski’s team wanted to make antibiotics that are initially inactive. Then a doctor could decide not only when but also where the antibiotics act in the body. Once an antibiotic has done its job, it could be turned off again with light.

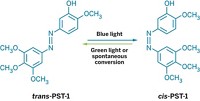
To make these on-off antibiotics, Szymanski modifies known drugs by adding azobenzene, a photoswitchable group. Ultraviolet light converts azobenzene from its trans form to its cis form. Blue light can reverse the process quickly. Otherwise, azobenzene will slowly recover its original, trans form over time. Szymanski adds the azobenzene to the drug scaffold so that the isomerization changes the molecule’s overall size, shape, or polarity, enabling it to bind its target in a microorganism.
So far, he says, it’s surprising how often just adding a photoswitch to an existing drug works. But, he notes, there is the potential to change the drug’s mode of action through this tinkering.
Glazer says such a photoswitchable approach is appealing but notes it has some limitations. One is the wavelength of light needed to do the switching. The best wavelengths to trigger isomerization are in the UV wavelength region. UV light is not very medically useful, Glazer says, “as the light does not penetrate more than a few millimeters into tissue and can induce DNA mutations” in healthy cells.
Red and infrared light, on the other hand, penetrate tissue deeply and safely.
At the end of last year, Szymanski and longtime collaborator Ben Feringa, one of the winners of the 2016 Nobel Prize in Chemistry, used that knowledge to take a step forward with photoswitchable antibiotics (J. Am. Chem. Soc. 2017, DOI: 10.1021/jacs.7b09281).
They took the molecular core of the antibiotic trimethoprim and added to it azobenzenes at different positions on the drug’s methoxyphenyl unit to create a library of UV-activated molecules. Screening against Escherichia coli, they found a molecule that was particularly potent when activated by UV light. To shift the wavelength at which this molecule’s trans-to-cis isomerization occurs, Szymanski’s team substituted atoms on the azobenzenes with fluorine and chlorine to alter the compounds’ activity. A chlorine-substituted version could be isomerized by red light. Not only that, but when this photoswitchable trimethoprim was switched on with red light, it was eight times as potent against E. coli as when it was switched off. To switch the drug off, the researchers used violet light, which rapidly returned the drug to its original, trans form.
Stefan Hecht at Humboldt University of Berlin points out that red and infrared light typically have their own limitations. “With UV light there’s a lot of energy that hits the molecule,” he says. “But if you irradiate a molecule with red or near-IR light, the amount of energy you dump into the molecule is very small,” he says. Basically, when red or IR light hits a photoswitching group, it does so with less energy, and thus the isomerization may be less likely than with UV light. That means the drug may be less potent.
But it is possible to overcome some of these limitations and also tune the molecules to longer wavelengths of light by focusing on designing better switches, Hecht says. This is what a number of groups are working on with azobenzenes, and as Szymanski and Feringa’s antibiotic—activated by red light while maintaining potency—proves, it can be successful.

Making photoswitchable anticancer drugs requires a different approach from one for designing antimicrobial compounds, says Oliver Thorn-Seshold from Ludwig Maximilian University Munich. With photoswitchable anticancer drugs, researchers need to ensure that the compounds bind to target proteins only in their “on” configuration. Binding in their “off” configuration would mean that the drugs could damage healthy cells. Photoswitchable antibiotics, on the other hand, target bacteria-specific proteins not present in human cells so they won’t damage human cells in either configuration.
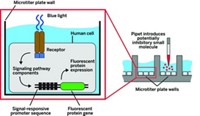
Thorn-Seshold is part of the development team at Munich-based CytoSwitch, which is led by Yelena Wainman and is designing compounds called photostatins, or PSTs (Cell 2015, DOI: 10.1016/j.cell.2015.06.049). When activated by blue light, these compounds, based on the structure of the natural product combretastatin A-4, interfere with cancer cell replication by targeting structural elements in cells called microtubules.
CytoSwitch’s photostatins, originally developed by Thorn-Seshold together with Dirk Trauner, who is now at New York University, are completely inactive before being hit with light, Thorn-Seshold says. That means that doctors could give patients high drug doses without concerns about adverse side effects. High doses also mean that low-intensity, biocompatible wavelengths of light can be used, because even if only a small fraction of the drug gets activated, the resulting levels of active molecule will be sufficient for treatment. Like other photoswitchable molecules, after being initially switched on with light, the photostatins eventually revert back to their original, inactive forms with a typical half-life of 15 minutes, Wainman says.
In developing these photostatins, Thorn-Seshold is running into a practical issue for the entire field—how to get light where it is needed to turn drugs on or off. He thinks that implanted light-emitting diodes could be a viable option. This is an existing technology, made mainstream by some tattooists who implant LEDs under the skin as a fashion statement. LEDs have also been used in optogenetics studies in mouse brains, where light is used to control neurons that have been genetically engineered to express light-activated ion channels.
Trauner, one of the pioneers of photopharmacology, doesn’t have to worry as much about light delivery with at least one of the projects he’s pursuing. He is working on restoring sight. “We are going for the low-hanging fruit,” Trauner says. Shining light into a person’s eyes won’t require any invasive LED implantation procedures, although some sort of visor may be needed.
Advertisement
Among the myriad of photoswitching compounds that Trauner is investigating is a compound that, when switched on in the eyes of blind mice, unblocks an ion channel and restores light sensitivity to the animals (Neuron 2012, DOI: 10.1016/j.neuron.2012.05.022). These mice were blind because the rods and cones in their eyes no longer worked, which also happens in human macular degeneration.
Trauner is confident that this vision-restoring work will become clinically relevant soon, or in the next few years. He says he now has around 120 examples of photoswitchable molecules working in cells or animals. Some of these are almost at the preclinical or clinical stage, he says. And he predicts that his vision-restoration products will be the first clinically approved photopharmacological drugs.
Still, getting light to areas of the human body other than the eyes for extended periods, such as days or weeks in the case of antibiotics, could present a significant problem for photopharmacology, says medicinal chemist Dennis Liotta of Emory University, who is editor in chief of ACS Medicinal Chemistry Letters. “Since light can’t easily penetrate deep into the skin of adults, one has to overcome the technical barrier of getting the light to the site of action,” he says. “There are various ways of doing this, but as far as I know, they all involve the use of sophisticated devices, and are all done in a hospital setting.”
The easiest places to reach are the skin and eyes because doctors can shine light directly on them. For harder-to-reach places like the gastrointestinal tract, lungs, or bladder, endoscopes could deliver light. For really hard-to-reach areas, including the brain and bone marrow, new strategies will have to be developed.
Trauner agrees with Thorn-Seshold that for many of these areas other than the eyes, implanted LEDs may ultimately be the most useful way to deliver light.
The field also faces possible regulatory hurdles before these photoswitchable drugs can reach the clinic. Szymanski says that each of these drugs is in fact two molecules—the “on” version and the “off” version—that need to be developed, tested for safety and toxicity, and submitted for approval to a regulatory body.
Hecht says medicinal chemists he talks to are often skeptical about how likely photoswitchable drugs are to make it through the process. “Making a small-molecule drug is already so complicated,” he says. Introducing the photoswitchable group could add complications that would make it harder to navigate the approval process.
Liotta thinks this could be less of a headache than it sounds. “The only issue I see is the toxicology profiles of the active and inactive compounds. If they are both safe, there shouldn’t be any intrinsic problems,” he says.
Both the U.S. Food & Drug Administration and the European Medicines Agency would not comment on the regulatory procedure for photoswitchable drugs because the field is still in its infancy.
Because of these regulatory considerations, Glazer says it is important to focus on the clinic and ensure the molecules that chemists are developing could lead to feasible treatments. This is important “so that time and resources are not expended pursuing ideas that are theoretically possible but in practice would be too cumbersome to be put into effect,” she says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter