Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Medicinal Chemistry
Synthesis of hindered dialkyl ethers shocked into new life
Rethinking an old electrochemical method boosts yields of the bulky compounds and lowers reaction times
by Leigh Krietsch Boerner
September 11, 2019
| A version of this story appeared in
Volume 97, Issue 36

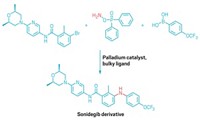
Hindered ethers are tricky little buggers. They’re important precursors for many products, especially in the synthesis of certain drugs. The bulkiness of the compounds helps prevent the body’s enzymes from metabolizing the drug molecule before it reaches its target. But this bulkiness also means the compounds are hard to make. Now Phil Baran of the Scripps Research Institute California and co-workers have resurrected an old electrochemical method to zap new life into hindered dialkyl ether synthesis, making 80 different ethers through zippier routes while also boosting yields (Nature 2019, DOI: 10.1038/s41586-019-1539-y).
Traditionally, chemists make ethers using the Williamson synthesis, which involves an oxygen nucleophile attacking a carbon center to kick off a leaving group through an SN2 type mechanism. In the case of hindered ethers, bulky groups on the attacking nucleophile get in the way and slow down the reaction and cut yields.
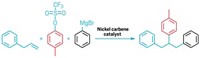
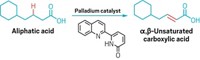
Baran’s team wanted to find a faster route to hindered ethers and decided to try the Hofer-Moest reaction. This electrocatalytic method was first published in 1902 and involves using an electric potential to drive the decarboxylation of a carboxylic acid, producing a carbocation that an alcohol nucleophile can then attack.
However, this reaction normally uses solvent quantities of the alcohol nucleophile, which can lead to generation of a lot of side products, such as alkynes from radical addition reactions, carbenes from elimination, and alcohols from hydration. Baran’s team could use less nucleophile and avoid side reactions by tweaking the electrode material and solvent used, as well as adding small amounts of sacrificial oxidants to prevent radicals in the solution from degrading the carbocation. The key was to make the carbocation long-lived so the small amounts of bulky nucleophile had time to react with it. In the previous version of the reaction, the carbocation “was living in a sea of nucleophiles waiting to attack it, like a steak thrown on a hoard of angry wolves,” Baran says. “In this case we basically had a carbocation sitting in a huge warehouse where in the corner maybe there was one puppy.”
The group ran over 1,000 reactions to optimize the reaction conditions and ended up making over 80 compounds (example shown). The new method had average yields around 43% and average reaction times of 9.8 hours. The older Williamson-based methods had average yields around 19% and took about 100 hours of reaction time. The team found that simple nucleophiles can also easily grab the carbocations, making hindered alcohols and alkyl fluorides, two other commercially important compounds. The method also tolerated a variety of more complex nucleophiles typically known for being sensitive to harsh reaction conditions, including some protected amines, acetals, and boronic esters.
The versatility of the reaction is really what’s important here, Baran says. For medicinal chemists, being able to apply similar methods to synthesize a variety of different molecules is essential. “They want to take modules of a thousand pieces of one thing and a thousand pieces of another thing and put them together and mix and match,” he says. “This method gives you the modularity.” For this work, Baran collaborated with industrial chemists from both Bristol-Myers Squibb and Pfizer.
“I think this is going to be something that pharmaceutical companies and medicinal chemists are going to pick up and use right away,” says Song Lin, an organic electrochemist at Cornell University. Baran and others, he says, have shown that chemists “can use the unique features of electrochemistry to power organic reactions.”
CORRECTION
This story was updated on Nov. 11, 2019, to correct Song Lin’s statement on Baran’s work.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter