Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Intramembrane Protease
Study suggests how protein cleavage in membranes might work
by Stu Borman
October 16, 2006
| A version of this story appeared in
Volume 84, Issue 42
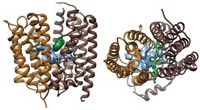
In a study that may be relevant to Alzheimer's and other diseases, researchers have obtained the first atomic-scale 3-D structure of an intramembrane protease and have proposed a molecular mechanism by which it might work.
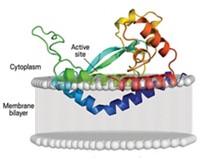
When cellular job assignments were handed out, intramembrane proteases got one of the hardest. These enzymes are embedded in membranes, which restricts their motion, and they cleave proteins at sites tucked within the membranes.
The enzymes are believed to be involved in many fundamental cellular processes. But researchers have been having a hard time imagining how intramembrane proteases might work. Water plays an essential role in reactions catalyzed by conventional proteases, but how would intramembrane proteases gain access to water inside membranes, which are hydrophobic? And how would substrates gain access to enzyme active sites in that environment?
Now, assistant professor of pharmacology Ya Ha and coworkers Yongcheng Wang and Yingjiu Zhang at Yale University have helped clarify the mechanism of such enzymes by determining and analyzing the structure of a bacterial intramembrane protease called GlpG (Nature, DOI: 10.1038/nature05255).
Cell biology group leader Matthew Freeman of the MRC Laboratory of Molecular Biology, in Cambridge, England, points out in a Nature commentary that this "landmark paper" reveals "a hydrophilic dent at the surface of the membrane, adjacent to the active site, ensuring that there is plenty of access for water to complete the peptide-breaking reaction." Ha says the position of residues conserved in enzymes of GlpG's class suggests a gating mechanism in which the residues move aside to permit the substrate to diffuse to the active site from the side. This proposal is still highly speculative, Ha adds.
Of three proteins implicated in Alzheimer's, two (presenilin-1 and -2) are intramembrane proteases, and the third ( -amyloid precursor protein) is their substrate. GlpG is not directly associated with Alzheimer's, but Ha says he hopes the new study will stimulate research leading to "a molecular explanation of the way mutations cause Alzheimer's disease and to rational drug design targeting presenilin-1 and -2."
-amyloid precursor protein) is their substrate. GlpG is not directly associated with Alzheimer's, but Ha says he hopes the new study will stimulate research leading to "a molecular explanation of the way mutations cause Alzheimer's disease and to rational drug design targeting presenilin-1 and -2."





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter