Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Hold That Thought
Slowly but surely, scientists are unveiling the complex chemical underpinnings of memory
by Sophie L. Rovner
September 3, 2007
| A version of this story appeared in
Volume 85, Issue 36

COVER STORY
Hold That Thought
WHEN WAS THE LAST TIME you misplaced your car in a parking lot? Gave you a bit of a scare, didn't it? What about when you blanked on the name of a longtime friend—did you wonder if you were showing the first signs of Alzheimer's disease?
These are not trivial fears. Memory is as vital to your trip to the grocery store as it is to your role at work and to your very personality.
To support these diverse responsibilities, memory is necessarily complex, and it has taken scientists more than a century to establish a basic understanding of its elaborate biochemical bases. Using isolated brain cells, brain scans, behavioral studies, pharmaceutical treatments, genetic engineering, and other tools, they have shown that memory is based on a series of biochemical events that induce changes in proteins in a network of the brain's neurons and that a lasting memory also requires structural changes in those neurons.
Researchers have discovered multiple genetic and pharmacological techniques to improve memory, an accomplishment that will no doubt capture the attention of an aging population (see page 22). They have also determined what happens when memory malfunctions. These findings could one day enable physicians to cure learning disabilities or to erase the unbearable memories associated with posttraumatic stress disorder.
But there's still plenty more to find out. The process of forgetting isn't well-understood, for instance. Nor do scientists have a firm grasp on how long-term memories can persist despite the rapid recycling of many of the molecules involved in their storage.
It's no wonder that the mechanism of memory is still something of a mystery. "The brain is the most complex machine in the whole world, and maybe the universe," says Nahum Sonenberg, a biochemist at McGill University, in Montreal.
It would be hard to dispute his assessment, given the mind-boggling statistics needed to describe this marvelous machine. The brain contains 100 billion neurons, each connected to thousands of other neurons via trillions of synapses.
"It's a very comforting thought that your brain has at least an order of magnitude more synapses than there are dollars in the U.S. debt," muses Gary S. Lynch, a professor of psychiatry and human behavior at the University of California, Irvine.
"That's a huge number of connections," adds Sonenberg. "And each time you see something, each time you learn something, you activate a different set of neurons" to form a unique memory of the event.
But how exactly is an event translated into a memory? Memory is constructed in stages. When the brain initially takes in an experience or a fact—such as a scene in a movie or an unfamiliar telephone number—the information first goes into short-term memory.
By definition, a short-term memory is transient, and the information it contains can be forgotten after a few hours or even just a few seconds. But if the information is freighted with sufficient weight—particularly if a person is paying close attention or if the information is associated with a stressful or emotional experience—it can be transferred into long-term memory, where it can endure for as long as a lifetime.
"If there's some event—a gunshot goes off or some wonderful event happens—the events surrounding that experience become crystalline in your memory, even if they precede that event," says Paul F. Worley, a neuroscientist at Johns Hopkins University. "The notion is that saliency—how important that information is to you—can distinguish whether that information is ultimately retained or not."
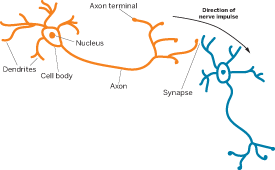
Different types of information are directed to different regions of the brain's cortex, the outer layer of the brain. Light given off by scenery in a movie, for instance, passes through the viewer's pupil and strikes rod and cone cells in the retina at the back of the eye. Pigment proteins in these cells absorb the light and convert its energy into a nerve impulse. Propagated by neurotransmitters and ion flows in and out of nerves, that impulse then shuttles along the optic nerve to the thalamus and then into the visual cortex at the back of the brain. Music from the sound track, on the other hand, passes from the ear through the thalamus and ends up in the auditory cortex in the middle of the outer sides of the brain.
MOST TYPES of information are next carried by nerve impulses into the hippocampus, which is located low in the middle of the brain. The hippocampus redirects information destined to form long-term memories back to the cortex. In the process, the hippocampus knits the associated memories together, creating links between the movie's scenery, music, story line, and so forth. When one aspect of the experience is later recalled, it will also call up the other associated memories in this neural network.
The transfer of information from cortex to hippocampus and back repeats until the memories become fixed. At that point, the hippocampus is no longer needed and the memories are lodged permanently in the cortex.
Some classes of information take another path in the brain as they transfer from short- to long-term storage. For instance, memory associated with skills and procedures—such as how to ride a bike—skips the hippocampus altogether when it's stored in the brain.
From a molecular standpoint, short- and long-term memories are stored in completely different ways. Formation of a short-term memory entails modification of preexisting proteins in neurons. But conversion of a short-term memory into a long-term memory—a process known as consolidation—requires gene expression and the synthesis of new proteins.
Most researchers believe that the neurons involved in short-term memory are the same ones that undergo consolidation. But some other researchers believe that short- and long-term memories require different types of neurons. Either way, consolidation, which stabilizes the new memory and makes it increasingly resistant to decay, might not be completed for days after the memory initially forms, says Alcino J. Silva, a neurobiologist at UCLA. Every time a memory is recalled, it has to be consolidated again or it will disappear.
The biochemical changes wrought during consolidation yield long-lasting increases in the strength of the synaptic connections between neurons and are associated with physical changes in the structure of the synapses. This enhanced connectivity, referred to as synaptic plasticity, is "thought to be the physiological mechanism underlying memory storage," notes neuroscientist Todd C. Sacktor of the State University of New York Downstate Medical Center, in Brooklyn.
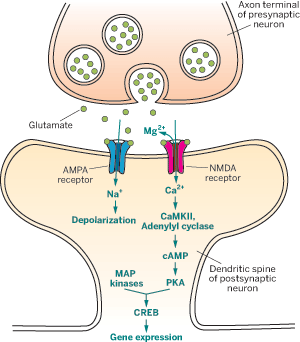
The formation of a memory involves a cascade of neuronal activations that start with neurons in the sensory organs and eventually involve neurons in the hippocampus and cortex, Silva explains. In this cascade, binding of the neurotransmitter glutamate to various receptors opens sodium and calcium ion channels.
The process begins when one neuron stimulates another by releasing glutamate into the synapse, or junction that links the two neurons together. After crossing the synapse, the glutamate molecules bind to α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors in the membrane of the postsynaptic neuron. The receptors then open ion channels that allow sodium ions into the negatively charged interior of the postsynaptic neuron. The sodium influx depolarizes the cell to some degree.
Depolarization affects the behavior of another group of postsynaptic glutamate receptors. These N-methyl-D-aspartate (NMDA) receptors control ion channels for calcium. Occasional release of glutamate from a single presynaptic neuron isn't sufficient to open these channels because they're plugged with magnesium ions. Under these circumstances, "neural activity wanes without inducing the biochemistry of memory formation," says Tim Tully, a neuroscience professor who studies memory at Cold Spring Harbor Laboratory, in New York.
If a presynaptic neuron strongly or repeatedly stimulates the AMPA receptors in the postsynaptic neuron with glutamate, however, the postsynaptic neuron depolarizes further. That pops the magnesium gatekeepers out of the ion channels associated with the NMDA receptors, allowing calcium ions to flow into the postsynaptic neuron.
The flow of calcium ions initiates a series of chemical events that lead to phosphorylation of the AMPA and NMDA receptors.
First, the calcium ions bind to and activate several enzymes in the postsynaptic neuron, including calmodulin. Ca2+/calmodulin activates other enzymes such as adenylyl cyclase and Ca2+/calmodulin-dependent protein kinase II (CaMKII). These two enzymes then phosphorylate and activate additional proteins. They also generate other compounds, including cyclic adenosine monophosphate (cAMP), which in turn boosts the activity of protein kinase A (PKA). Both PKA and CaMKII phosphorylate AMPA and NMDA receptors.
The modified receptors are more efficient at passing sodium and calcium ions into the neuron and thus increase its response to glutamate. Additional AMPA receptors held in readiness elsewhere in the neuron insert in the postsynaptic membrane to enhance synaptic strength further.
Moreover, PKA and mitogen-activated protein (MAP) kinases activate cAMP response element-binding protein (CREB), a transcription factor, which increases the synthesis of new AMPA receptors. CREB also boosts production of other proteins important for memory consolidation, including structural proteins such as actin that serve as the basis for the formation of enlarged or new synapses between the neurons. Numerous other proteins are involved in the postsynaptic neuron's response to glutamate.
After the nerve impulse passes, the postsynaptic receptors release their bound glutamate molecules, which then diffuse away or are reabsorbed by the presynaptic neuron. The postsynaptic neuron reverts to its "resting state" by expelling sodium ions and adjusting other ion concentrations to reestablish a negatively charged interior.
In sum, a strong or repeated burst of glutamate from one or more presynaptic neurons induces a greater sensitivity in the postsynaptic neuron to subsequent glutamate exposure. This "long-term potentiation" can persist for hours, weeks, or—some scientists believe—even years. The glutamate burst also alters the structure of existing synapses. Together, these changes strengthen the synaptic connections between the neurons.

If glutamate produces a strong enough response or excitation in a neuron, the nerve impulse can pass along to the next neuron and the next, and thus propagate through memory structures such as the hippocampus. Given sufficient signal strength, the impulse can move beyond the hippocampus and back into the cortex. The enduring memory that results is distributed over a network of neurons whose connections have been reinforced through long-term potentiation and growth of additional synapses. The memory can be recalled when the network is reactivated, though very little is known about this process.
BUT WHAT happens to memories that last for a while but aren't permanently retained?
Understanding of the process of forgetting is murky, and the proposed mechanisms are controversial. Some researchers believe certain parts of the brain employ "long-term depression" to clean the slate, so to speak. In the hippocampus, for example, long-term depression weakens connections between synapses that are only rarely stimulated. Under these conditions, glutamate binding to NMDA receptors on the postsynaptic membrane brings only a small number of calcium ions into the neuron. This small amount of calcium activates enzymes that dephosphorylate the receptors, making them less responsive to glutamate.
Long-term depression also reduces the number of AMPA receptors in the postsynaptic membrane. It also might alter the structure of synaptic connections between the neurons. The net result of these steps is to return the neuron to a state in which it's ready to receive new information.
Memory is even more complex than this summary indicates, and any magazine piece about the subject can give only a flavor of the rapidly growing body of knowledge about the intricate and nuanced biochemical mechanisms involved.
For instance, although NMDA receptors must be activated for memory to work, their overactivation can be neurotoxic, says Graham L. Collingridge, a neuroscientist at the University of Bristol, in England. "To function in memory, NMDA receptors let calcium into neurons. But that calcium signal then has to be reduced back down to baseline levels. If it's elevated for any prolonged period of time, it can start activating proteases and so forth, which can start destroying the cell." That mechanism might explain what happens in Alzheimer's patients and might even occur in normal age-related memory loss, Collingridge believes. "There's a lot of work going on right now to try and work this out," he says.
Advertisement
Curiously, DNA methylation, a technique the body uses to permanently switch off unneeded genes, might also be used transiently during memory storage. J. David Sweatt, a neurobiologist at the University of Alabama, Birmingham, reported on this process earlier this year (Neuron 2007, 53, 857). He found that training regimens that led to the formation of fear-related memories increased methylation, and hence deactivation, of a memory-suppressor gene in rats. The training also demethylated and activated a memory-promoting gene. Conversely, blocking DNA methylation interfered with the ability of the rats to form fear-related memories. The modifications were all reversible, suggesting that changes in DNA methylation in the adult nervous system aren't necessarily permanent.
Other groups have shown that memory formation triggers transient acetylation, phosphorylation, or phosphoacetylation of the histone proteins around which DNA is wrapped, says Craig M. Powell, a neurologist at the University of Texas Southwestern Medical Center. These so-called chromatin modifications help regulate gene expression. He suggests that these short-term modifications might be "involved in the consolidation of memories but are not necessarily the substrate for the memory itself."
In fact, Powell says, "one big question that remains is, 'What is the self-regenerating biochemical change at synapses that underlies long-lasting synaptic plasticity and learning and memory?' " Most proteins are recycled in days or even minutes, so what maintains the synaptic changes that last for years?
SUNY's Sacktor appears to have discovered one of those proteins. He has shown that protein kinase M zeta (PKMζ)—a long-lasting fragment of protein kinase C (PKC)—is required to maintain long-term potentiation and to hang on to long-term memories.
Long-lasting synaptic changes might also be preserved through the formation of durable new slots for the additional AMPA receptors that are inserted in the postsynaptic membrane during long-term potentiation, Powell says. The slots might be composed of specific postsynaptic scaffolding molecules that secure the receptors within the membrane. Whereas the receptors and the individual scaffolding molecules are constantly recycled, the slots themselves somehow maintain a place for the receptors in the membrane, he adds.
ANOTHER MYSTERY centers on the way a neuron responds to activation of some but not all of its synapses.
A neuron "has to have a program for saying, 'Let's double the number of AMPA receptors or glutamate receptors at this particular synapse,' " Powell notes. "The problem is, that would be easy to do cellwide. Except that's not how synaptic plasticity works. The changes don't happen all throughout the neuron. They happen only at the points of synaptic contact that were activated in a certain way. You can't just make more 'thought proteins' and put them everywhere."
In some cases, production of certain proteins is restricted to specific regions within a neuron that's involved in processing a memory. For instance, the gene for PKMζ is transcribed into messenger RNA in the neuron's nucleus. The mRNA then commutes to the neuron's dendrites, the branched structures that receive incoming signals from other neurons. Dendrites are decorated with dendritic spines, small protrusions that form one half of a synapse. The PKMζ mRNA churns out the protein only at dendrites whose synapses have been activated through long-term potentiation. The protein maintains an increase in the number of AMPA receptors at those potentiated synapses.
Likewise, messenger RNA for Arc/Arg3.1 shuttles from the nucleus to the neuron's dendrites to produce the protein locally at potentiated synapses. Last year, Johns Hopkins' Worley and several other researchers published multiple papers describing the protein's function in long-term memory, including its role in regulating the number of AMPA receptors at a given synapse (Neuron 2006, 52, 403, 437, 445, 461, and 475).
But neurons can apparently call on other methods to accomplish changes only at potentiated synapses. For example, some researchers believe that synapses that are strengthened during the formation of a memory are "tagged molecularly" to capture and utilize necessary proteins that were produced elsewhere in the neuron, Powell says. "No one knows the nature of this tag," but there's electrophysiological evidence to suggest that it exists, he adds. "And so when you have new proteins synthesized, they're targeted to these specific tagged synapses, and somehow they can solidify these molecular events—such as AMPA receptor insertion or synaptic structural changes—and make them long-lasting."
Recent advances in both genetic techniques and imaging technology are enabling scientists to delve ever deeper into the physiological changes that accompany memory formation. Using a sophisticated new technique known as restorative deconvolution microscopy, Lynch recently showed that a learning experience enlarged individual synapses involved in long-term potentiation by about 50% in rats. Synapses that weren't affected didn't change size (J. Neurosci. 2007, 27, 8031).
"The idea that connections between brain cells would change with learning" goes back to the late 1800s, Lynch says. "For us to visualize this process after all these years is a big thrill. There's good reason for thinking that bigger synapses have more transmitter receptors and are functionally more potent, so it means that we now know what is the encoding device of memory."
One of the great questions in behavioral neuroscience centers on where a particular memory is stored in the brain, Lynch adds. "With the methods we've developed, we can now go through the entire cortex of an animal and map the sites at which a memory has been deposited." The physical and biochemical changes that make up such a "memory trace" are referred to as an "engram."
It's possible that in disorders such as mental retardation or depression, or even in aging, memory deficits arise because memories aren't deposited in their normal location. Lynch's technique will allow scientists to explore such issues in animal models of these conditions.
But what determines where a memory is stored? UCLA's Silva recently used confocal microscopy to determine that levels of the transcription factor CREB dictate which of the eligible neurons in the brain are engaged to store a particular memory. He found that memories are preferentially channeled into neurons that contain a higher level of CREB (Science 2007, 316, 457).
Memory has turned out to be a whole lot more complex than expected. "People used to look for the memory molecule," Powell says. "They wanted to find out what's the one biochemical change at a synapse that changes its strength. But it's much more complicated than that." And though the contradictory needs of ephemeral and enduring memories sometimes seem irreconcilable, memory researchers refuse to be daunted. "We tend to grasp onto the things that we do understand and try to build little stories out of those," Powell says. "It's a very complicated cell biological problem. That's why it's so much fun, and that's why there are thousands of labs working on these issues."


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter