Advertisement
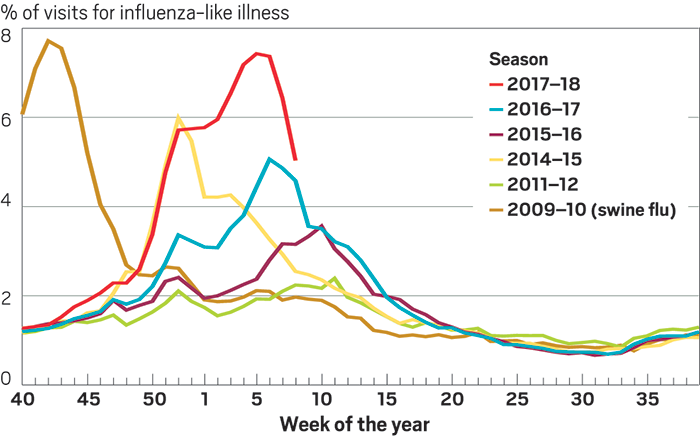
By all accounts, the 2017–18 influenza season has been a bad one. It feels like everyone’s family has been hit, sometimes twice. Schools have had to shut down to disinfect; hospitals are overcrowded.
In brief
Hoping to thwart influenza infections, many vigilant people roll up their sleeves for an annual flu shot. But this preventive measure doesn’t come close to providing the same protection that people are used to from other vaccinations. For years, scientists have been working to make a universal flu vaccine that’s effective against all possible strains of the virus and will last for years. Read on to learn about their progress.
More
By February, the number of people in the U.S. going to see their doctor with flu-like symptoms reached levels not seen since 2009, when the country battled the swine flu pandemic. As February drew to a close, a time when the seasonal virus usually starts to wind down, the Centers for Disease Control & Prevention (CDC) said 45 states were still reporting widespread flu activity. Flu-related deaths can be tough to pin down, but in a bad season like this one, as many as 650,000 people worldwide die from flu and flu-related complications, according the to the World Health Organization.
Advertisement
“Almost everything we’re looking at is bad news,” said CDC Acting Director Anne Schuchat during a press briefing on Feb. 9. “It’s a wake-up call about how severe influenza can be and why we can never let down our guards against this virus,” she said.
A major reason this flu season has been so rough is that the vaccine just wasn’t that good. Preliminary CDC estimates find the vaccine has been 36% effective overall, meaning the vaccine reduced the risk of getting sick and having to go to the doctor by 36%. What’s more, the component of the seasonal vaccine that fights the H3N2 strain of flu—the one making most people sick this season—has had an effectiveness of just 25%, according to CDC.
“If you look at the efficacy of seasonal flu vaccines over years, it’s striking that in the very best year you get 60% efficacy and in the worst year, your efficacy can be as low as 0–10%,” says Anthony S. Fauci, director of the National Institute of Allergy & Infectious Diseases (NIAID), which is part of the National Institutes of Health. “That’s completely unacceptable when you compare it to the efficacy of vaccines that we’ve made successfully over the years for other infections.”
The measles vaccine, for example, is 98% effective; the polio vaccine is more than 95% effective. When it comes to flu vaccines, Fauci says, “We’ve got to do better.”
Vaccines protect against viruses by making the body do all the hard work. Inject a little inactivated virus and the body will recognize it as an invader, sending T cells to fight the virus and B cells to create antibodies that block it from infecting cells. Once the body has mounted this defense, certain T and B cells remember the infection and surge into action when they encounter the live virus.
Related: Toward A Universal Flu Shot
One challenge with creating a good flu vaccine is forecasting. Experts analyze influenza data from around the world to figure out which strains are most likely to make people sick in the upcoming season. But there is a lag of a year or more between when data are collected and when people get a flu shot. To some extent, it’s a guessing game. Sometimes the experts guess wrong.
Another issue is that flu viruses can make small mutations that render the vaccine less effective—a phenomenon known as antigenic drift. Sometimes an entirely new version of the virus can move from pigs or birds into people. Experts describe this as a shift, and this is what happened during the 2009 swine flu pandemic, which CDC estimates made over 60 million people in the U.S. sick.
Fauci and many other flu experts argue what’s really needed is a so-called universal flu vaccine. Ideally, this kind of immunization would provide protection against all flu strains and would last for years, maybe even for someone’s entire lifetime. The tricky ways the virus mutates make it a tough proposition. But around 20 companies and government agencies are chipping away at it, using a range of strategies and technologies to create broadly protective flu vaccines. Some are even being tested in the clinic.
“It turns out to be a pretty challenging goal,” says Jesse Bloom, who studies the molecular evolution of viruses at Fred Hutchinson Cancer Research Center.
The problem begins with flu’s ability to change faster than most other infectious diseases. Bloom explains that the flu virus can make small tweaks to the two main proteins on its surface: hemagglutinin and neuraminidase. Sticking out of the virus like pins in a pincushion, these proteins are the H and N in scientists’ shorthand for different strains of flu, like H1N1 or H3N2. Hemagglutinin’s job is to get the virus into cells where it can replicate. Neuraminidase cuts the virus free from cells so it can recirculate and go on to infect more cells.
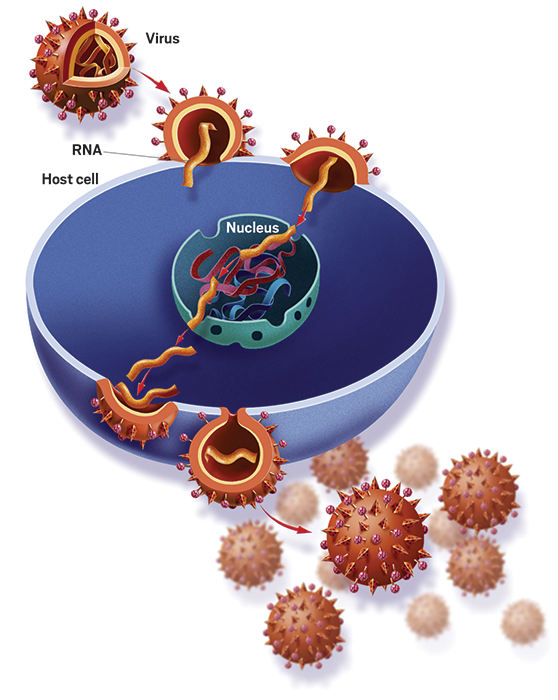
“One of the main ways our immune system can protect us against the virus is to make antibodies that bind to the hemagglutinin protein,” Bloom says. Those antibodies most easily latch onto the part of the protein that’s furthest away from the virus—or the head of that pin-shaped structure. “That’s also the part of the hemagglutinin that changes most when the virus evolves from year to year,” he says.
“The goal of a universal vaccine is to somehow reengineer the vaccine so that it induces our body to make antibodies against parts of the hemagglutinin protein that do not change very rapidly in nature,” Bloom explains. This includes the part of hemagglutinin that connects the head to the larger virus particle—researchers call it the stem—as well as certain conserved portions on the head.
“It’s conceptually simple but technically difficult,” says Neil King, a biochemistry professor at the University of Washington who designs nanomaterials for use in potential universal flu vaccines. Scientists know it’s possible to stop a lot of different flu strains with one antibody—they’ve shown such antibodies exist. But what they can’t figure out is how to prompt the body to make them. King explains that these so-called broadly neutralizing antibodies have strange features that are hard for our immune system to make.
“The biggest challenge is the fact that the virus has so much capacity to change and still be an influenza virus,” says Barney Graham, deputy director of NIAID’s Vaccine Research Center. His team is trying to convince the body to create antibodies to hemagglutinin’s stem by creating ferritin nanoparticles—essentially little protein balls—that display only headless stems. The nanoparticles display the headless stems in a symmetric array that the body recognizes as viruslike and will mount a defense against. In animal studies, NIAID researchers have shown that inoculating with nanoparticles that have a stem from an H1 flu can protect against an H5 flu strain that’s lethal to unvaccinated animals.
Flu by the numbers
Annual influenza epidemics cause roughly
3 million to 5 million
of severe illness worldwide.
290,000 to 650,000
people die from the flu each year.
Typical flu incubation period:
2 days
People with the flu ar most contagious in the first
3 to 4 days
after their illness begins, but some healthy adults may be able to infect others beginning 1 day before symptoms develop and up to 5–7 days after becoming sick.
Overall effectiveness of the 2017–18 flu vaccine for all age groups combined:
36%
Breakdown:
H1N1 component:
67%
effective
H3N2 component:
25%
effective
Influenza B components:
42%
effective
Sources: World Health Organization, Centers for Disease Control & Prevention
By using stems from the two main groups of influenza A, NIAID scientists think they can protect against most influenza A viruses, Graham says. This strategy could even work against viruses that have drifted and possibly ones that represent a pandemic shift. He says they are hoping to start clinical trials of the nanoparticles later this year.
Researchers at Icahn School of Medicine at Mount Sinai are also making progress with a vaccine candidate that targets hemagglutinin’s stem. Led by Peter Palese, Adolfo García-Sastre, and Florian Krammer, the team has developed a virus particle that displays the hemagglutinin stems from H1 with heads from influenza strains that circulate only in birds.
First they inoculate with one stem and head combination; then they inoculate with the same stem but a different head. The idea is that this one-two punch trains the body to recognize and develop antibodies against the stem regardless of its head. Two vaccine candidates made using this approach, one that can be injected and one that can be inhaled in a mist form, are in Phase I clinical trials, sponsored by GlaxoSmithKline and the Bill & Melinda Gates Foundation, respectively.
“It’s too early to say whether this will work sufficiently well and be long lasting enough to replace seasonal vaccines,” Palese says.
Scientists at Inovio Pharmaceuticals are making a universal vaccine candidate that tackles hemagglutinin in a different way. Whereas the NIAID and Mount Sinai teams are trying to coax the immune system into recognizing certain parts of the flu virus, Inovio’s synthetic DNA vaccine prompts our cells into making hemagglutinin itself—which the immune system then recognizes and makes antibodies against. To do this, Inovio’s vaccine candidate includes DNA instructions for making a few different hemagglutinin proteins. These instructions are so-called consensus DNA, in which each amino acid has been chosen because it is the one most commonly found in a particular position in dozens or even hundreds of hemagglutinins.
The approach allows the vaccine candidate to cover a broader section of the genetic map, says Inovio CEO J. Joseph Kim. In collaboration with scientists from Wistar Institute and the Public Health Agency of Canada, Inovio recently reported its vaccine candidate protected ferrets (commonly used as test animals for influenza) from all the pandemic H1N1 viruses from the past 100 years. That, Kim says, indicates “there’s a high probability that we will be able to protect against all potential shifts in the next 100 years.”
Related: Flu Vaccine Race Against The Clock
Although Inovio has reported work with only H1N1 strains of flu, Kim says the company has also made vaccine candidates in the same manner for H3N2 strains and influenza B strains. “When you combine all of these into a single administration, you will indeed have a universal flu vaccine that will potentially protect against all known and unknown strains,” he says.
Targeting hemagglutinin isn’t the only approach scientists are taking to create a universal flu vaccine. Vaccitech, a spin-out from the University of Oxford, is targeting conserved proteins that are located inside the virus rather than on the surface. Vaccitech’s vaccine candidate uses a virus that can’t replicate in humans to deliver two conserved flu proteins. When the body sees these proteins, it boosts production of influenza-specific T cells. Rather than block infection, these immune cells kill any cells the virus has already infected.
Vaccitech CEO Thomas Evans points out that because the technology goes after conserved virus proteins, it doesn’t have to keep up with the ever-evolving virus. “So the vaccine I make this year is good 10 years from now,” he says.
Vaccitech’s vaccine candidate, known as MVA-NP+M1, is in Phase II clinical trials in combination with the seasonal vaccine. The current trial investigates whether the combination offers better protection against the flu than the seasonal vaccine on its own in people over age 65.
Although Vaccitech is developing its candidate as a combination therapy, Evans thinks it could be used as a stand-alone vaccine for pandemic strains of influenza A (it’s only partially effective against influenza B). “We do not know how long our vaccine will work, but what we do know is that the T cells that we generate last for about two years,” Evans says.
BiondVax Pharmaceuticals also has a universal influenza vaccine candidate, called M-001, in Phase II clinical trials. M-001 is a synthetic protein that combines peptides from two of influenza’s conserved proteins and hemagglutinin. Like Vaccitech’s candidate, M-001 stimulates T cells to fight influenza. It also spurs B cells to action by prompting them to make flu-fighting antibodies. At the end of 2017, the company announced it was planning a Phase III trial in Eastern Europe for the 2018–19 flu season.
Should any of these approaches prove effective, there are still caveats. NIAID’s Graham points out that when the body’s B cells make flu antibodies, they often produce antibodies to the first virus or vaccine they ever encountered, even if that’s not what they’re currently exposed to. It’s a phenomenon researchers call original antigenic sin. This can make it tough to effectively vaccinate adults for new strains of flu. If scientists do find a way of creating a universal flu vaccine, Graham says, “It may have to be started in very young children to be the most effective.”
Even with the progress scientists are making toward a universal vaccine, it will be years before such preventive measures are part of the flu-fighting arsenal. Until then, flu researchers say, the seasonal vaccine is still the best defense.
