Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Water
William Tarpeh
Waste wizard is turning what we normally flush away into a resource
by Michael Torrice
August 25, 2019
| A version of this story appeared in
Volume 97, Issue 33


Vitals
Current affiliation: Stanford University
Age: 29
PhD alma mater: University of California, Berkeley
If I were an element, I would be: “Tungsten. I always liked that its symbol was so unexpected from its name and that it’s a critical part of light bulbs.”
I’ve overcome adversity in the lab by: “I’ve experienced my share of impostor syndrome and noninclusive environments, and it has helped me to intentionally design an inclusive, collaborative atmosphere in my lab group, classrooms, and any room I enter.”
Must-have in the lab: “Slack for lab communication. Shout-out to my students for teaching me the wonders of Slack!”
Must-have on the road: “Noise-canceling headphones.”
People don’t normally look at what they flush down the toilet and see something worth recovering. But William Tarpeh does.
The Stanford University environmental and chemical engineer is developing ways to extract resources from human waste, such as nitrogen in urine, to make valuable products, like fertilizers and cleaning products. This idea of looking at waste as a resource could help us meet the environmental challenges of a growing human population and changing climate, he says.
Our urine is a concentrated solution of nutrients, Tarpeh points out. It contains nitrogen, phosphorus, and potassium, all crucial elements found in fertilizers. But normal wastewater treatment strategies often end up dumping those nutrients into waterways, where the nitrogen and phosphorus can feed algal blooms that choke aquatic ecosystems, leading to oxygen-deprived dead zones. Meanwhile, industrial methods for making nitrogen fertilizers require massive amounts of energy and emit metric tons of greenhouse gases. So pulling the nitrogen out of our urine could combat several environmental problems, Tarpeh says.
He also thinks it could help improve sanitation and access to clean water in the developing world. “If we can turn human waste into a valuable product, then we can generate revenue from toilets,” he says. “And then toilets in areas like sub-Saharan Africa and Southeast Asia become a revenue-generation center instead of a cost sink.”
Tarpeh has developed multiple methods for extracting nitrogen from urine. In graduate school at the University of California, Berkeley, he developed ion exchange resins that can bind the ammonium in urine. He envisions people inserting cartridges filled with these resins into toilet pipes. Special collection services would then pick up the cartridges, treat them to extract the captured ammonium, and then send them back to consumers.
Another method Tarpeh is working on is called electrochemical stripping, which first involves separating the positively charged species in urine from the negatively charged ones. As a result of this electrochemical process, pH increases around the positively charged ions, converting ammonium to ammonia, which can then diffuse through a gas-selective membrane, away from the other cations. Ammonia then reaches an acid trap, where the gas converts back to ammonium for collection.
“William is stepping outside of dogma and thinking more broadly,” says Krista Wigginton, one of his postdoc advisers at the University of Michigan–Ann Arbor. For example, she says, environmental engineers tend to think about centralizing treatment of municipal wastewater. “William’s PhD work flipped that idea upside down.”
Also, Tarpeh doesn’t focus on just the science behind his ideas; he’s also concerned about their implementation in real-world settings, says Kara Nelson, his graduate adviser at UC Berkeley. For example, he brings his methods out into the field, including in resource-limited areas in Africa, to test them.
Tarpeh hopes his work will help tackle an enormous problem that humanity faces in the 21st century: scarcity. “We don’t have enough to go around, and so we have to develop methods to really squeeze the most value out of every chemical,” he says. “We have to figure out what is the maximum number of times we can use a molecule before we deem it as waste.”
People don’t normally look at what they flush down the toilet and see something worth recovering. But William Tarpeh does.
The Stanford University environmental and chemical engineer is developing ways to extract resources from human waste, such as nitrogen in urine, to make valuable products, like fertilizers and cleaning products. This idea of looking at waste as a resource could help us meet the environmental challenges of a growing human population and changing climate, he says.
Advertisement
Our urine is a concentrated solution of nutrients, Tarpeh points out. It contains nitrogen, phosphorus, and potassium, all crucial elements found in fertilizers. But normal wastewater treatment strategies often end up dumping those nutrients into waterways, where the nitrogen and phosphorus can feed algal blooms that choke aquatic ecosystems, leading to oxygen-deprived dead zones. Meanwhile, industrial methods for making nitrogen fertilizers require massive amounts of energy and emit metric tons of greenhouse gases. So pulling the nitrogen out of our urine could combat several environmental problems, Tarpeh says.
He also thinks it could help improve sanitation and access to clean water in the developing world. “If we can turn human waste into a valuable product, then we can generate revenue from toilets,” he says. “And then toilets in areas like sub-Saharan Africa and Southeast Asia become a revenue-generation center instead of a cost sink.”
Tarpeh has developed multiple methods for extracting nitrogen from urine. In graduate school at the University of California, Berkeley, he developed ion exchange resins that can bind the ammonium in urine. He envisions people inserting cartridges filled with these resins into toilet pipes. Special collection services would then pick up the cartridges, treat them to extract the captured ammonium, and then send them back to consumers.
Another method Tarpeh is working on is called electrochemical stripping, which first involves separating the positively charged species in urine from the negatively charged ones. As a result of this electrochemical process, pH increases around the positively charged ions, converting ammonium to ammonia, which can then diffuse through a gas-selective membrane, away from the other cations. Ammonia then reaches an acid trap, where the gas converts back to ammonium for collection.
“William is stepping outside of dogma and thinking more broadly,” says Krista Wigginton, one of his postdoc advisers at the University of Michigan–Ann Arbor. For example, she says, environmental engineers tend to think about centralizing treatment of municipal wastewater. “William’s PhD work flipped that idea upside down.”
Also, Tarpeh doesn’t focus on just the science behind his ideas; he’s also concerned about their implementation in real-world settings, says Kara Nelson, his graduate adviser at UC Berkeley. For example, he brings his methods out into the field, including in resource-limited areas in Africa, to test them.
Tarpeh hopes his work will help tackle an enormous problem that humanity faces in the 21st century: scarcity. “We don’t have enough to go around, and so we have to develop methods to really squeeze the most value out of every chemical,” he says. “We have to figure out what is the maximum number of times we can use a molecule before we deem it as waste.”
Vitals
Current affiliation: Stanford University
Age: 29
PhD alma mater: University of California, Berkeley
If I were an element, I would be: “Tungsten. I always liked that its symbol was so unexpected from its name and that it’s a critical part of light bulbs.”
I’ve overcome adversity in the lab by: “I’ve experienced my share of impostor syndrome and noninclusive environments, and it has helped me to intentionally design an inclusive, collaborative atmosphere in my lab group, classrooms, and any room I enter.”
Must-have in the lab: “Slack for lab communication. Shout-out to my students for teaching me the wonders of Slack!”
Must-have on the road: “Noise-canceling headphones.”
Research at a glance
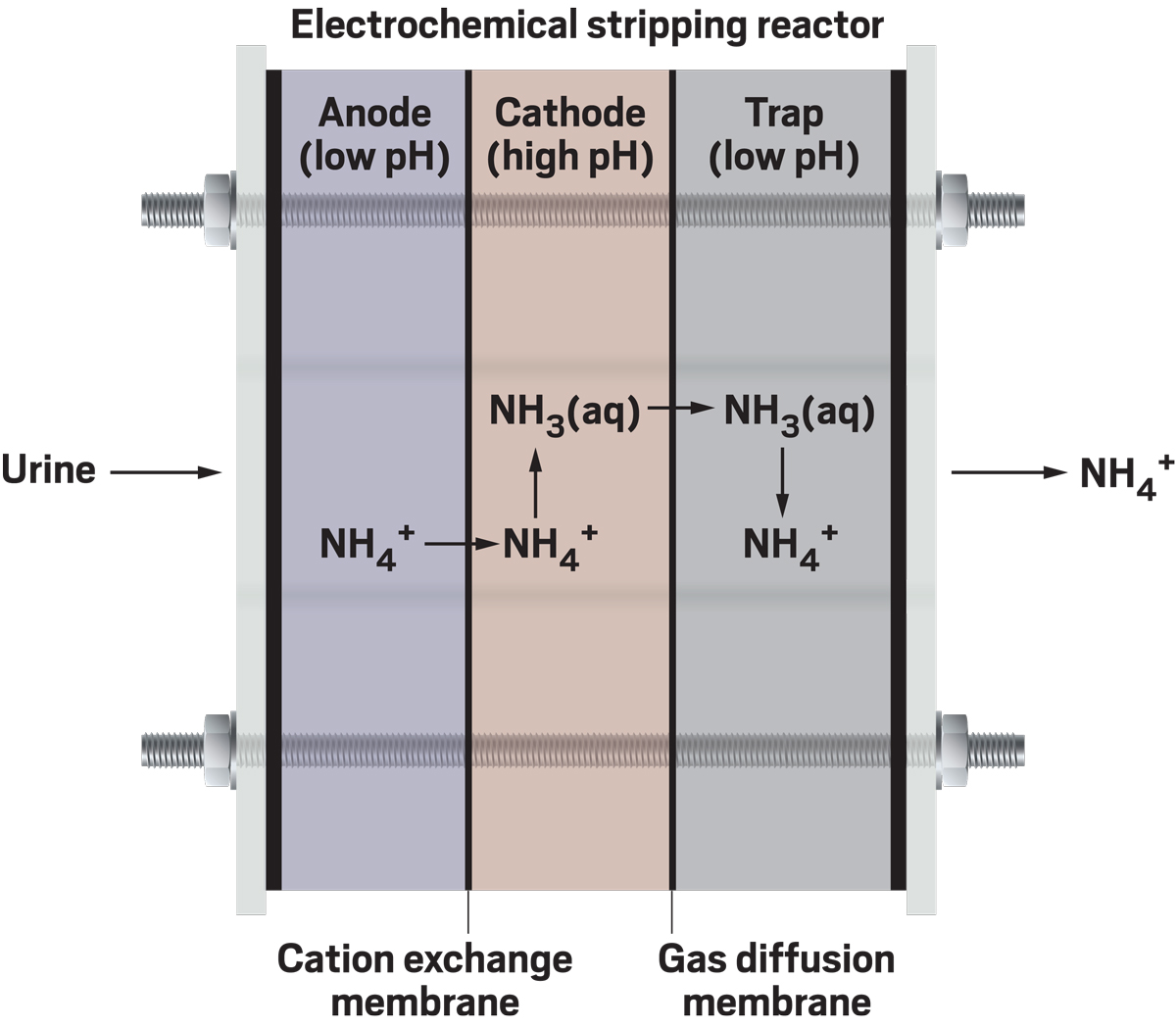
Credit: Adapted from Environ. Sci. Technol./Yang H. Ku/C&EN
Tarpeh built a reactor that isolates ammonium from urine by first separating it and other positively charged ions from negatively charged ones. As a result of this process, the pH increases, converting the ammonium to ammonia, which then diffuses into an acid trap, where it becomes ammonium again.
Three key papers
“Electrochemical stripping to recover nitrogen from source-separated urine”
(Environ. Sci. Technol. 2018, DOI: 10.1021/acs.est.7b05488)
“Comparing ion exchange adsorbents for nitrogen recovery from source-separated urine”
(Environ. Sci. Technol. 2017, DOI: 10.1021/acs.est.6b05816)
“Sanitation For Low-Income Regions: A Cross-Disciplinary Review”
(Annu. Rev. Environ. Resour. 2019, DOI: 10.1146/annurev-environ-101718-033327)




















Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter