Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
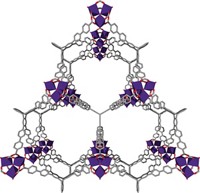
Metal-Organic Frameworks
Conducting charge is MOFs’ new trick
This unexpected property of the porous materials is opening doors to new applications, like charge storage devices, electrochemical sensors, and electrocatalysts
by Mitch Jacoby
November 28, 2021
| A version of this story appeared in
Volume 99, Issue 43

In brief
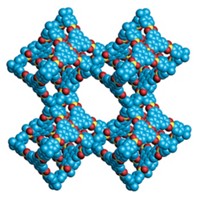
Metal-organic frameworks (MOFs) are mostly pockets of empty space within a honeycomb structure of organic linkers and metal ions. Most scientists might assume such materials are incapable of conducting charge, but surprisingly, some MOFs do it well. That little-known, unexpected property, which scientists are trying to understand, is pushing MOFs into new territory. A small but quickly growing number of researchers is exploiting the rare combination of porosity, high surface area, and charge conductivity in these materials to create electronic sensors, charge storage devices, electrocatalysts, and other applications traditionally outside MOFs’ purview.
For 25 years, chemists have been finding applications that take advantage of the extreme surface areas of metal-organic frameworks (MOFs). In just 1 g of some of these porous materials, the areas can reach nearly 8,000 m2—larger than a standard soccer pitch. That’s far greater than the surface area packed inside zeolites, activated carbon, and other porous materials that chemists rely on to catalyze reactions and capture molecules.
The record-setting surface area and porosity of MOFs provide an enormous number of sites for molecules to dock and react and plenty of room for them to come and go. These features make MOFs ideal for mediating catalytic reactions as well as for storing, separating, and purifying gases. For example, chemists have designed MOFs for industrial carbon capture, for conveniently storing clean-burning transportation fuels, and for pulling water out of thin air.
But some chemists have realized that MOFs have another trick up their sleeve—they can conduct charge. That relatively unknown feature could be a game changer because it opens the door to using MOFs in entirely different types of applications, such as charge storage materials, electronic chemical sensors, and electrocatalysts. Researchers are just starting to explore conductive MOFs and how they transport charge. But chemists are already testing the materials in new applications that take advantage of MOFs’ newly appreciated conductivity trick.
The rise of conductive MOFs
Conductive MOFs aren’t new, but they’ve flown under the radar until recently. Ten years ago, hardly any studies had been published on the subject. In 2019, researchers published more than 125 papers. And the number keeps climbing. Part of the attraction to these materials is that they boast seemingly contradictory properties.
“Almost by definition, porosity and electrical conductivity are at odds with each other,” says Mircea Dincă, a materials chemist at the Massachusetts Institute of Technology who has been working on conductive MOFs for a decade (Chem. Rev. 2020, DOI: 10.1021/acs.chemrev.9b00766).

“We think of electric charge traveling through condensed phases,” Dincă says, referring to solids and liquids. “But MOFs are mostly emptiness, mostly nothingness. How can you make electric current travel through space that is mostly empty? That’s counterintuitive,” he says. Yet it’s possible with conductive MOFs.
MOFs are crystalline compounds composed of metal ions or clusters of ions joined by organic linker units. They form honeycomb-like structures filled with an array of molecule-sized voids. Studies of conductive MOFs have revealed that the materials can conduct charge through multiple mechanisms. One of the common mechanisms, known as the through-bond pathway, involves charge moving through a network of coordination bonds between metal centers and functional groups in the organic linkers. Iron-based MOFs containing azolate linkers, which are heterocycles with nitrogen atoms, exhibit some of the highest conductivities in this category.
Another mechanism, known as the extended conjugation pathway, also involves bonds between the metal atoms and the linkers. But in this pathway, the organic linkers contain functional groups, such as diamines and dithiols, that are conjugated with the linker’s carbon core.
Other pathways feature charge transport between adjacent molecular layers in a MOF. This so-called through-space pathway is common in MOFs with naphthalenes, anthracenes, and other aromatic groups. Each molecular layer of these MOFs stacks like playing cards via interactions between the π bonds in the MOF’s aromatic rings. In general, the closer the 2D layers, the higher the conductivity of these materials.
But conduction pathways aren’t always limited to a MOF’s metal ions and organic linkers. Sometimes MOFs can conduct charge via electroactive molecules that bind within the material’s pores.

“Understanding how MOFs conduct charge is a curiosity-driven question—a fundamental science issue,” Dincă says. Studying these pathways can help chemists boost MOF conductivity and understand design principles for making MOFs that perform well in charge-based applications. MOFs’ modular design enables chemists to use their giant tool box of synthetic methods to fine-tune MOF properties and tailor each material for a target application. For example, by swapping one set of linkers for another, chemists can modify structural features such as pore size and connectivity. They also can tweak the chemical environment inside a MOF’s pores by judiciously selecting the metal and pairing it with organic linkers endowed with certain functional groups.
“Because MOFs self-assemble from well-defined molecular precursors, you can dial in structural features and chemical properties with atomic precision,” says Katherine A. Mirica, a Dartmouth College chemist who specializes in framework compounds. That level of control also extends to tuning MOF conductivity, providing chemists with a powerful way of exploring the various charge-transport mechanisms.
No matter how a MOF conducts charge, chemists are excited to exploit this new feature of the materials. They think that MOFs’ enormous surface areas could allow the materials to outperform those already used in some applications.

For example, an electronic sensor that has thousands of additional sites per square centimeter to bind target molecules could be more sensitive than one using non-MOF materials. Or a similar increase in catalytically active sites on an electrode could be leveraged “to get a lot more oomph out of a fuel cell,” Dincă says.
Exploring new applications
As research on conductive MOFs moves into high gear, chemists continue refining ways to prepare the materials, analyzing and boosting charge transport, and developing inventive applications.
MOFs by the numbers
~8,000 m2
Surface area per gram of material of a record-setting MOF
~7,100 m2
Surface area of a standard soccer pitch
~3,000 m2
Surface area per gram of material of leading types of activated carbon
~3 vs. >125
Number of publications on conductive MOFs in 2009 and 2019
>200
Number of MOFs for which a charge-transport mechanism has been studied
Sources: Chem. Rev.; Angew. Chem., Int. Ed.
One conductive-MOF application that’s sure to leave others in the dust and has found commercial interest is energy storage. Dincă’s group has been working with engineers at Automobili Lamborghini, a manufacturer of high-end sports cars, to develop MOF-based supercapacitors to power its futuristic, all-electric concept muscle car, Terzo Millennio.
Why not use lithium-ion batteries like the ones in a Tesla or other consumer electric cars? “Because to accelerate quickly, muscle cars need a lot more power than luxury cars,” Dincă says, “and today’s batteries just can’t cut it.”
Batteries can store a lot of energy but can’t unload it quickly. Supercapacitors typically cannot store much energy, but they can release what they have in a flash, repeatedly and at high frequency, which is good for the rapid acceleration that muscle cars need. To work well, supercapacitors require a charge storage material that is both highly porous and highly conductive. Very few materials available today fit the bill, Dincă says. One that does is activated carbon, which is currently used in various forms in commercial supercapacitors. But Dincă and coworkers wanted to make something that works even better.
By fine-tuning a family of MOFs featuring nickel and cobalt ions and organosulfur linkers, the MIT-Lamborghini team has come up with a highly porous material that exposes more surface area to electric charge than is possible with traditional supercapacitor materials. Supercapacitors made from the new material—for which the team has filed for patent protection—have double the energy density of today’s state-of-the-art devices, providing a major boost in energy storage and power delivery.
Chemical sensors might not be as flashy as Italian sports cars, but they have a wider range of applications, including detecting toxic gases or investigating physiological processes that produce those gases. At Dartmouth, Mirica’s group recently used conductive MOFs to develop chemiresistive sensors for ammonia, hydrogen sulfide, and nitric oxide. These devices detect analytes by sensing a change in electrical resistance when the molecules bind to the MOFs. The sensors achieved part-per-million level sensitivity, which is good for gas sensors in personal safety equipment, Mirica says, but not good enough to use in biological studies of how these gases are produced.
So her team designed new sensors using a conductive MOF featuring two different groups to bind the gas molecules: a copper-oxygen-based group and a nickel phthalocyanine unit. The team reported that the improved sensor can detect the three gases selectively at the part-per-billion level (J. Am. Chem. Soc. 2019, DOI: 10.1021/jacs.8b11257).
Describing the study as a promising proof of concept, Mirica says commercialization of these sensors would require rigorous evaluation of their stability and chemical selectivity to ensure that they can reproducibly differentiate among a large number of analytes in real-world mixtures.
In related work, the Dartmouth team used tailor-made 2D MOFs added to a carbon electrode to build a neurochemical detector. The researchers found that by using a nickel triphenylene MOF, they could selectively detect dopamine and serotonin at nanomolar concentrations—a clinically relevant level—in simulated urine solutions. Other electrochemical methods for achieving that level of sensitivity are more costly and time consuming (J. Am. Chem. Soc. 2020, DOI: 10.1021/jacs.9b13402).

At the Korea Advanced Institute of Science and Technology, Il-Doo Kim, Chungseong Park, and coworkers modified a MOF to boost its conductivity and make it capable of detecting a gas. Basically, they dexterously deposited metal nanoparticles in the MOF’s pores. The team treated a copper triphenylene MOF with metal salts, then reduced the precursors, forming confined platinum-ruthenium particles measuring just 1.5 nm in diameter. The group showed that the decorated MOF could be used as a chemiresistive sensor to detect nitrogen dioxide at the sub-parts-per-million level (Adv. Mater. 2021, DOI: 10.1002/adma.202101216).
Meanwhile, at the Technical University of Dresden, Xinliang Feng and coworkers have been working on electrocatalytic applications for conductive MOFs. They showed that a 2D conductive MOF could mediate the catalytic electroreduction of carbon dioxide to carbon monoxide. That conversion has been widely studied as a way to make synthesis gas, a mixture of CO and hydrogen that is a starting material for synthesizing fuels and chemicals. The MOF catalyst, which features copper-phthalocyanine and zinc-bis(dihydroxy) units, performed well, producing CO with 88% selectivity and remaining stable during a test run exceeding 10 h (Nat. Commun. 2020, DOI: 10.1038/s41467-020-15141-y).
Almost unknown just 10 years ago, conductive MOFs are now starting to take off. “Given the structural and chemical diversity of MOFs and their design versatility, it is likely that the potential of MOFs for electrocatalysis and other applications has not yet been fully exploited,” says Song Jin, a materials chemist at the University of Wisconsin–Madison.
“But instead of taking an Edisonian approach and trying out every material for every possible application, people should carefully consider the most rational strategies and debate the important scientific questions,” Jin says. For example, researchers should continue to explore charge-transport mechanisms and methods for boosting MOF conductivity. And they should focus on making cost-effective materials that are designed to be chemically stable, durable, and scalable.
That approach, Jin says, “could accelerate the discovery process and lead to killer applications for MOFs.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter