Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Drug Discovery
Covid-19
The antivirals that weren’t: drug repurposing for COVID-19 produced misleading results
A common class of compounds disrupts the cell rather than stops the virus
by Leigh Krietsch Boerner
June 29, 2021
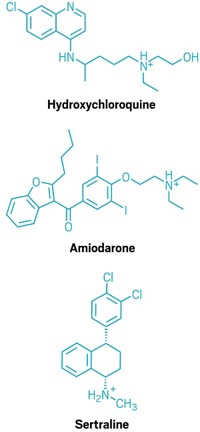
At the start of the COVID-19 pandemic in early 2020, scientists shifted into high gear to find a treatment. One of the quickest routes was to study drugs that treat other diseases. But some drugs that looked like they had antiviral activity against SARS-CoV-2 are not active against the virus that causes COVID-19, according to a new study (Science 2021, DOI: 10.1126/science.abi4708). A large, multi-institutional team led by Nevan J. Krogan, Kevan M. Shokat, and Brian K. Shoichet from the University of California, San Francisco, tested 23 repurposed cationic amphiphilic drugs (CADs), including hydroxychloroquine, amiodarone, and sertraline. The researchers found that what looked like antiviral activity in cells was actually the result of a mechanism called phospholipidosis, which disrupts cells by interrupting fundamental cellular processes.

Support nonprofit science journalism
C&EN has made this story and all of its coverage of the coronavirus epidemic freely available during the outbreak to keep the public informed. To support us:
Donate Join Subscribe
“These drugs should basically be dropped,” Shoichet says. “They do not have a future as antiviral, anti-COVID-19 drugs.” Antiviral compounds use various strategies to keep viruses from replicating in a cell. For example, they can prevent the production of key enzymes. Phospholipidosis harms infected cells, but it’s not therapy, Shoichet says. The team studied the CADs in animals, which became sick from phospholipidosis before the drugs killed infected cells. “Even at the very highest concentrations, we didn’t see an antiviral effect,” he says.
Phospholipidosis, a buildup of fatty molecules outside the cell membrane, happens when organelles called lysosomes can no longer properly break down or distribute proteins to the rest of the body. The process can cause serious damage, including lung and liver injury, although sometimes it has no side effects, says Jayme Dahlin, preclinical medical director at the National Center for Advancing Translational Sciences, part of the National Institutes of Health. “It’s really hard to study,” says Dahlin, whose research focuses on compounds that cause off-target toxicity.
Studying approved drugs is a quick way to find a treatment for a new disease because their safety and use in humans has already been well established. Since the start of the pandemic, scientists have initiated hundreds of clinical trials of repurposed molecules for COVID-19, and Shoichet emphasizes that his group’s findings do not mean other classes of drugs won’t work against the disease. But he thinks researchers should stop investing time and money in CADs. “More broadly, this whole class is now under a dark cloud as a treatment for COVID-19,” Shoichet says. “My instinct would be to drop them off or certainly to look very carefully and make sure you know that the antiviral activities that are seen are not correlated with phospholipidosis,” he says.
In the past 15 months, Shoichet, Krogan, Shokat, and coworkers have published a number of papers on repurposing drugs and drug candidates for COVID-19. In particular, the researchers had zeroed in on compounds that bind to σ receptors as possible antivirals. But after digging further, “we eventually came to realize that there was no real relationship between binding to the σ receptors and activity on the virus,” Shoichet says. Some drugs that bind strongly to the σ receptors were mediocre or even ineffective against SARS-CoV-2, while others that don’t work well on the σ receptor were effective at killing the virus, he says. This lack of correlation between potency on the target and activity on the virus was deeply disturbing, Shoichet says. “Because it says your whole hypothesis might be wrong.”
And it was. By looking carefully at these σ-receptor molecules and integrating what they knew about pharmacology and medicinal chemistry, the researchers started to identify a pattern. The molecules that seemed best at killing the virus “were these cationic, greasy drugs, like amiodarone,” Shoichet says. Amiodarone is the poster child for acting through phospholipidosis, he says. “There looked like there was a correlation between our most potent drugs and molecules that were known to do this,” Shoichet says.
The researchers also ran mouse studies of the four compounds that were the most potent against SARS-CoV-2 in their earlier cell studies—amiodarone, sertraline, PB28, and tamoxifen. That research confirmed that with those drugs, phospholipidosis, not antiviral activity, was responsible for the harm to the infected cells. They gave the mice varying amounts of the drugs and then infected them with SARS-CoV-2. The team measured how much virus had replicated in the mice’s lungs after a 3-day incubation period. They saw little to no antiviral effect. In addition, the researchers treated cells with the 23 CADs and saw lipid blobs in whorled patterns in the cells’ vacuoles, which indicate phospholipidosis. “It’s like the cells developed a bad case of lipid acne,” Shoichet says.
The researchers searched DrugBank Online, a database that contains information about clinical trials worldwide, to understand how many CADs had been tested in COVID-19. They found that 316 studies using CADs as antivirals advanced all the way to Phase 3 clinical trials. Nearly 60% of those involved either hydroxychloroquine or chloroquine. The researchers estimated that these clinical trials might have cost over $6 billion in the past year.
When you test thousands of drugs, a lot of compounds are going to seem promising, NCATS’s Dahlin says. “Lesson learned the hard way: most of the things that look interesting in these early studies turn out to be not useful in the long run,” he says. They’re either false positives, or they’re stopping the virus in a way that’s not useful for an animal or human.
The findings could have wide-reaching implications. The mechanism behind these false positives “is probably not just for COVID-19,” Shoichet says. I think it’s going to be true for antiviral drug discovery in general.” He adds that “this paper does not sink all drug-repurposing efforts. But it says, ‘Here’s a way to distinguish wheat from chaff,’ ” he says. Some important compounds can come out of drug-repurposing approaches. And the assays that the researchers did in this paper give scientists a straightforward way to screen for these false positives. Now, Shoichet says, “you can get rid of them quickly and focus on the ones that are real.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter