Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Catalysis
Chemists shine light on new way to think about reductive elimination
Computational study confirms alternative mechanism for C-C bond forming reaction
by Leigh Krietsch Boerner
November 4, 2020
| A version of this story appeared in
Volume 98, Issue 43

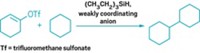
Reductive elimination reactions, in which a transition metal catalyzes new bond formation between two molecules, are one of organic chemists’ most powerful tools. For example, one flavor of these reactions, palladium-catalyzed allylic alkylations can make new C–C bonds while tolerating many functional groups. In addition, these mild reactions are one of the few ways chemists can generate compounds containing hindered quaternary carbons.
Through a detailed computational study, Brian Stoltz and coworkers at the California Institute of Technology have figured out that some Pd-catalyzed reductive eliminations go through a different mechanism than previously thought, providing a new understanding of how reductive eliminations work (J. Am. Chem. Soc., 2020, DOI: 10.1021/jacs.0c09575).

Traditionally, chemists thought the mechanism involved transition states featuring bond breaking and forming between three key atoms. Stoltz and coworkers studied a specific Pd-catalyzed reductive elimination reaction in which the desired C-C bond forms within the same molecule (shown). The chemists determined that the transition state for this reaction involves bond forming and breaking between seven atoms. Describing this seven-centered reductive elimination process allows chemists to expand the way they think about this fundamental reaction, Stoltz says. This mechanism is not the way chemists learn how reductive eliminations work in graduate school, he says, but it suggests that there are other ways that reductive elimination can happen. “It could change the way people think about what’s possible.”
For this computational study, the researchers focused solely on the Pd-centered transition state. Generally, this transition state is difficult for chemists to study experimentally because it’s not the rate-limiting step of the reaction, so it exists briefly. Chemists have suspected that this transition state involves 7 key atoms, and is organized in a ring, like those found in pericyclic reactions. Not only did Stoltz and coworkers confirm this suspicion, but also they showed that the Pd’s d-orbitals allow the transition state to be aromatic, which means it’s stabilized through extended conjugation of orbitals across the seven atoms. This stabilization allows the transition-state energy to be low enough for the reaction to proceed. In addition, they found that because of the geometry of Pd’s d orbitals this aromaticity doesn’t fit the traditional structure, instead it is shaped like a Möbius strip, which is a connected loop with one surface and a half turn.
The researchers also used a type of chemical bonding theory to explain the mechanism of the reaction in terms of arrow pushing. This drawing method in which arrows depict where electrons move in a chemical reaction is the way that many organic chemists are taught to understand these transformations.
Through detailed analysis, the researchers showed the inner workings of an important organometallic reaction, says Dean Tantillo, a computational organic chemist at the University of California, Davis. They used hardcore quantum chemistry to build a model using familiar, intuitive concepts, he says.
This study “allows us to think about some of the things we can improve on,” in the current reaction, and gives a high-level theoretical backing for the feasibility of new kinds of reaction, Stoltz says.
UPDATE:
This story was updated on Nov. 6, 2020, to clarify how the discussed reaction is similar to pericyclic reactions, and to add Brian Stoltz and team's university affiliation.





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter