Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.

Mónica Pérez-Temprano
Intermediates interrogator wants to improve old reactions and find new ones
by Carmen Drahl
August 20, 2018 | APPEARED IN VOLUME 96, ISSUE 33
The bullying that Mónica Pérez-Temprano experienced as a teenager made school a nightmare. "People thought that I didn't fit in," she says. Now, as one of the youngest research group leaders in Spain, she wears being different like a badge of honor.
While most chemists wait until a reaction works before trying to understand its steps in detail, Pérez-Temprano advocates probing the mechanisms of transition-metal catalysts alongside reaction development—especially when things aren't yet running smoothly. When something's not working, it means a reaction intermediate failed to form efficiently, she explains. Stabilizing that intermediate could make a transformation more efficient or point to new reactivity.
Since starting her lab at the Institute of Chemical Research of Catalonia, Pérez-Temprano has used cobalt as a test case for the theory that reactivity of intermediates can guide reaction design. Cobalt and other first-row transition metals are less expensive and more sustainable than the precious metals, such as palladium, that chemists typically use to forge new bonds. Moreover, catalysts containing first-row transition metals can potentially yield intermediates—and thus products—that catalysts containing precious metals cannot.
That variety would be a boon to chemists synthesizing materials or drug candidates. Yet reaction development for first-row metals is in relative infancy because mechanisms are unclear, making processes tough to steer in a desired direction.
Fortunately, "Mónica is laser focused on mechanism," says University of Michigan, Ann Arbor's Melanie Sanford, her postdoctoral adviser. "It was pretty brave of her to tackle these cobalt reactions," Sanford adds, because those intermediates are notorious for falling apart easily.
Undaunted, Pérez-Temprano's team last year captured an elusive seven-membered-ring cobalt intermediate for the first time. She's excited to harness this intermediate for some cool cobalt chemistry, but she also wants to spread the word that focusing on intermediates could solve all sorts of chemical problems.
Ever mindful of her experience growing up, she takes pride in training undergraduates and advocating for women in science so they can forge their own unique paths. "I've found my place in the world," she says. "And I've learned that mentoring and being visible changes other people's lives."
Watch Pérez-Temprano speak at the American Chemical Society national meeting on Aug. 20 in Boston.
Vitals
Current affiliation: Institute of Chemical Research of Catalonia
Age: 35
Ph.D. alma mater: University of Valladolid
Role models: My previous advisers, Pablo Espinet and Juan Casares at the University of Valladolid, and Melanie Sanford at the University of Michigan, Ann Arbor. They have been such an incredible inspiration for my career, not only as researchers but also as great mentors.
If I were an element, I would be: Cobalt. Cobalt is a chameleon. Depending on its environment, it can potentially behave like almost any other metal.
Latest TV show binge-watched:"House of Cards"
Walk-up song: "Machine" by MisterWives
Research at a glance
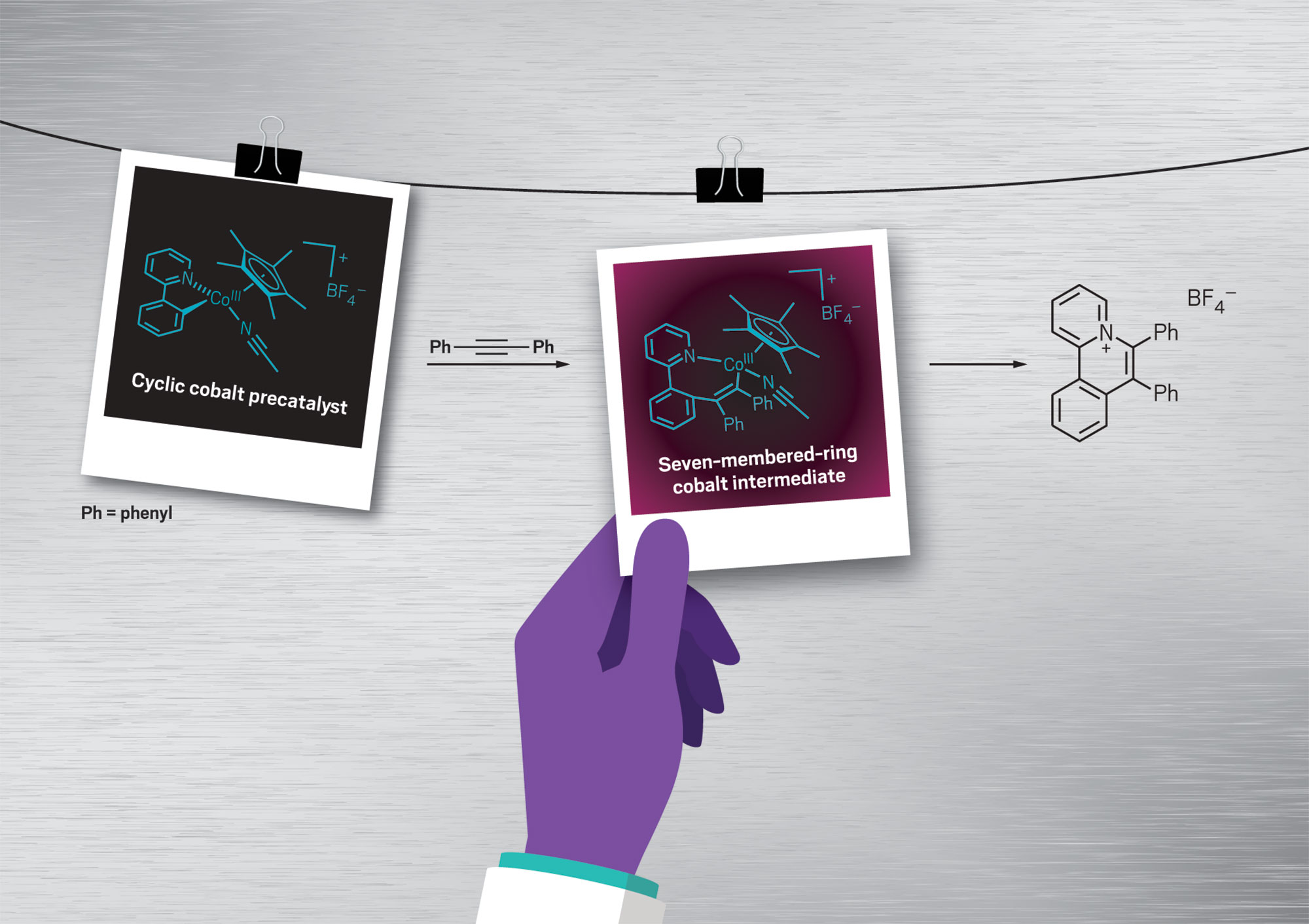
Credit: Adapted from Angew. Chem./Will Ludwig/C&EN/Shutterstock
Pérez-Temprano's team aims to capture snapshots of fleeting molecular species while developing new reactions, using the structures as "knowledge building blocks" for designing better catalysts. The researchers trapped and characterized both a cyclic cobalt precatalyst and an elusive seven-membered-ring cobalt intermediate.
Three key papers
"Competition between sp3-C–N vs sp3-C–F Reductive Elimination from PdIV Complexes"
(J. Am. Chem. Soc. 2014, DOI: 10.1021/ja411433f)
Catalysis
Amazing Women
Mónica Pérez-Temprano
Intermediates interrogator wants to improve old reactions and find new ones
by Carmen Drahl
August 19, 2018
| A version of this story appeared in
Volume 96, Issue 33


Vitals
Current affiliation: Institute of Chemical Research of Catalonia
Age: 35
Ph.D. alma mater: University of Valladolid
Role models: My previous advisers, Pablo Espinet and Juan Casares at the University of Valladolid, and Melanie Sanford at the University of Michigan, Ann Arbor. They have been such an incredible inspiration for my career, not only as researchers but also as great mentors.
If I were an element, I would be: Cobalt. Cobalt is a chameleon. Depending on its environment, it can potentially behave like almost any other metal.
Latest TV show binge-watched:“House of Cards”
Walk-up song:“Machine” by MisterWives
Three key papers
“Competition between sp3-C–N vs sp3-C–F Reductive Elimination from PdIV Complexes” (J. Am. Chem. Soc. 2014, DOI: 10.1021/ja411433f)
“Capturing Elusive Cobaltacycle Intermediates: A Real-Time Snapshot of the Cp*CoIII-Catalyzed Oxidative Alkyne Annulation” (Angew. Chem. Int. Ed. 2017, DOI: 10.1002/anie.201704744)
“HFIP-Assisted C–H Functionalization by Cp*CoIII: Access to Key Reactive Cobaltacycles and Implication in Catalysis” (Angew. Chem. Int. Ed. 2018, DOI: 10.1002/anie.201806847).
The bullying that Mónica Pérez-Temprano experienced as a teenager made school a nightmare. “People thought that I didn’t fit in,” she says. Now, as one of the youngest research group leaders in Spain, she wears being different like a badge of honor.
While most chemists wait until a reaction works before trying to understand its steps in detail, Pérez-Temprano advocates probing the mechanisms of transition-metal catalysts alongside reaction development—especially when things aren’t yet running smoothly. When something’s not working, it means a reaction intermediate failed to form efficiently, she explains. Stabilizing that intermediate could make a transformation more efficient or point to new reactivity.
Since starting her lab at the Institute of Chemical Research of Catalonia, Pérez-Temprano has used cobalt as a test case for the theory that reactivity of intermediates can guide reaction design. Cobalt and other first-row transition metals are less expensive and more sustainable than the precious metals, such as palladium, that chemists typically use to forge new bonds. Moreover, catalysts containing first-row transition metals can potentially yield intermediates—and thus products—that catalysts containing precious metals cannot.
That variety would be a boon to chemists synthesizing materials or drug candidates. Yet reaction development for first-row metals is in relative infancy because mechanisms are unclear, making processes tough to steer in a desired direction.
Fortunately, “Mónica is laser focused on mechanism,” says University of Michigan, Ann Arbor’s Melanie Sanford, her postdoctoral adviser. “It was pretty brave of her to tackle these cobalt reactions,” Sanford adds, because those intermediates are notorious for falling apart easily.
Undaunted, Pérez-Temprano’s team last year captured an elusive seven-membered-ring cobalt intermediate for the first time. She’s excited to harness this intermediate for some cool cobalt chemistry, but she also wants to spread the word that focusing on intermediates could solve all sorts of chemical problems.
Ever mindful of her experience growing up, she takes pride in training undergraduates and advocating for women in science so they can forge their own unique paths. “I’ve found my place in the world,” she says. “And I’ve learned that mentoring and being visible changes other people’s lives.”—.


















Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter