Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Microscopy
Cryo-electron microscopy reaches resolution milestone
Detailed protein structures are fueling drug-discovery efforts
by Mark Peplow, special to C&EN
September 27, 2020
| A version of this story appeared in
Volume 98, Issue 37

“It’s like a miracle compared to a few years ago,” says Bridget Carragher, codirector of the Simons Electron Microscopy Center at the New York Structural Biology Center, a research consortium. “It’s quite astonishing.”
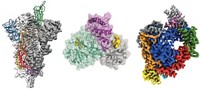
Carragher is describing the most detailed portrait to date of a GABAA receptor, a cell membrane protein that is an important target for many drugs. Back in 2014, the atoms in the first crystal structure of a GABAA receptor were hidden within lumpy, elongated blobs. In the new image, many of those atoms now stand out as distinct individuals.
It reveals not just the receptor’s detailed form, but also exactly how histamine—a molecule that targets the receptor—nestles snugly in one of the protein’s pockets. The positions of the surrounding atoms show how hydrogen bonds reach from different parts of the protein to grip one of the histamine nitrogen atoms. Water molecules fill the rest of the pocket, swaddling histamine like bubble wrap. The image is one of the sharpest pictures of a protein-ligand embrace ever created by cryogenic electron microscopy (cryo-EM), revealing features as small as 1.7 Å—approaching the 1 Å size of a single hydrogen atom.
Cryo-EM has been around for decades, but in the past 10 years it has surged forward to become a powerhouse tool for investigating the structures of proteins and other large biomolecules. Indeed, three cryo-EM pioneers—Jacques Dubochet of the University of Lausanne, Joachim Frank of Columbia University, and Richard Henderson of the MRC Laboratory of Molecular Biology (LMB)—scooped up the 2017 Nobel Prize in Chemistry for their work.
The GABAA receptor structure, reported in May, vividly illustrates how much detail the method now provides (bioRxiv 2020, DOI: 10.1101/2020.05.22.110189). The same paper, which was published on a preprint server and therefore has not yet been peer reviewed, also contained a structure of apoferritin, a highly symmetric protein often used as a benchmark to push cryo-EM to its limits, at a record-breaking resolution of 1.2 Å. In this structure it is possible to pick out individual hydrogen atoms, the first time that cryo-EM has achieved fully atomic resolution in a protein. A second team reported its own 1.25 Å apoferritin structure on the same day (bioRxiv 2020, DOI: 10.1101/2020.05.21.106740).
By enabling researchers to better see how small molecules bind to protein active sites, as with histamine to the GABAA receptor, the technique’s improved resolution is starting to assist the structure-based design of potential drugs—including treatments for COVID-19.
Yet cryo-EM still faces some major hurdles. Sample preparation is often troublesome, and it takes many hours to collect enough data for a decent structure. Cryo-EM is also eye-wateringly expensive: almost all the best structures rely on Titan Krios microscopes, 4 m tall behemoths that come with a price tag of over $6 million and swallow thousands more every day in running costs. So even as researchers celebrate the field’s latest resolution record, they are already working on ways to make cryo-EM faster, easier, and cheaper, hoping that it can become a more accessible analytical tool.
Sharper imagery
X-ray crystallography, which produces diffraction patterns from X-rays ricocheting off atoms in crystallized proteins, is still the dominant technique used to visualize protein structures. When it works, it still tends to deliver protein structures at higher resolution than cryo-EM. But some proteins stubbornly refuse to form the requisite crystals, while others, like the GABAA receptor, are too floppy to produce especially sharp images using X-rays.
Cryo-EM offers an alternative way to wrangle such recalcitrant proteins. Instead of crystallizing proteins, researchers flash freeze them into thin disks of ice using liquid ethane. The instrument shoots a beam of electrons into the disks, and the electrons scatter off the protein atoms, projecting a pattern onto a detector. Multiple snapshots—thousands, or even hundreds of thousands—capture the patterns from every conceivable orientation. Software crunches through the resulting terabytes of data to reconstruct a 3-D model of the protein.
Cryo-EM offers other advantages, too. While researchers often modify proteins with reagents to help crystallize them for X-ray analysis, cryo-EM can tackle proteins in more natural states. The technique also generally requires much less protein. And since many proteins carry out their biological tasks by changing shape, taking snapshots of a protein that has been flash frozen in different conformations can uncover important details about how it functions—something that a single, static X-ray image cannot do.
Various improvements in cryo-EM, including better detectors and refined software, have helped the technique to advance in leaps and bounds. To achieve their record-breaking apoferritin structure, A. Radu Aricescu and Sjors H. W. Scheres at the LMB worked with microscope manufacturer Thermo Fisher Scientific to build an instrument with top-shelf components that squeeze as much data as possible from every electron.
This is just the tip of a broader trend. Since 2010, the average resolution of a cryo-EM structure has improved from 15 Å to about 6 Å, and it is increasingly common for cryo-EM to deliver protein structures in the range of 3–4 Å. “The resolution has been getting better and better, on more difficult systems,” says Harren Jhoti, CEO of Astex Pharmaceuticals, a drug-discovery company based in Cambridge, England.
Better sample preparation has also helped. One persistent challenge has been that as researchers flash freeze a protein sample, some fraction of the molecules become damaged or adopt a weird conformation. The more such wonky molecules pollute a sample, the longer the instrument needs to make a good image. And in some cases, these distortions render the sample useless.
Five years ago, Carragher developed an automated sample preparation spray process that helps samples solidify much more quickly—wicking liquid away via a specialized sample support—thereby reducing protein damage. Last year, the system was commercialized by SPT Labtech in the UK.
At the LMB, Christopher J. Russo is using graphene to help safeguard samples. His team has created gold grids with an atom-thin layer of graphene, modified with functional groups such as amines, thiols, and carboxylic acids. These bind to proteins, preventing them from bouncing around during sample preparation and holding them steady as the image is acquired (Proc. Natl. Acad. Sci. U.S.A. 2019, DOI: 10.1073/pnas.1904766116).
Vaccine action
Further improvements to parts such as electron detectors might allow 1 Å cryo-EM protein structures in the next few years, Scheres says, but pushing any further might be all pain with no gain. “All the chemistry stops at 1 Å resolution, and the biology mostly stops at 3 Å,” Russo says. After all, while the images produced by high-resolution cryo-EM are breathtaking to behold, the real power comes from using these detailed views to understand how the proteins function and to develop new drugs to interact with them.
For example, at 3 Å, the primary and secondary structures of a protein are clearly visible, which is enough to answer many important questions in structural biology. Pfizer, for example, relied on cryo-EM to develop a vaccine for respiratory syncytial virus (RSV). It is the most frequent cause of serious respiratory tract infections in infants and young children and is responsible for 120,000 deaths every year. “Cryo-EM was quite instrumental to that project,” says Seungil Han, head of Pfizer’s cryo-EM facility in Groton, Connecticut.
Atoms in sight

To create the vaccine, the company mimicked a so-called fusion glycoprotein on the surface of the virus that helps it traverse cell membranes and cause infection. Researchers created 360 stabilized forms of this fusion protein and screened them for the potential to stimulate a protective antibody response. Pfizer used cryo-EM to study how candidate proteins from this group interacted with antibodies, to help it find the ideal form to act as a vaccine. This fusion-protein vaccine entered Phase III clinical trials earlier this year.
Many drug candidates, however, are based on the chemistry of small molecules, not whole proteins. This is where sub-2 Å resolution could make a difference, allowing researchers to conduct structure-based design of better drugs.
Astex, for example, is beginning to use cryo-EM to screen small organic molecules for their ability to bind to proteins. Screening a library of a few thousand of these molecules can reveal which functional groups and structural features produce the strongest interactions with target proteins. Piecing together several of these molecular units can then provide a promising drug candidate, a strategy known as fragment-based drug design.
Astex has long used automated X-ray crystallography to carry out these screens, but it has just invested in its own Krios microscope, anticipating that cryo-EM could help it investigate a much wider range of proteins. In a proof-of-principle study published last year, the company screened cocktails of fragments against a target protein for cancer chemotherapy called pyruvate kinase 2 (Drug Discovery Today 2019, DOI: 10.1016/j.drudis.2019.12.006). “It’s clearly much slower than we can do it with X-ray, but this is just a start,” Jhoti says.
Battling a pandemic
Cryo-EM is proving to be a vital tool in understanding SARS-CoV-2, the virus that causes COVID-19. Earlier this year, researchers used the method to quickly determine structures of the virus’s spike protein, which helps it enter human cells, as well as key polymerase and protease enzymes. The spike protein is a key target in efforts to develop vaccines against the virus. A detailed 2.5 Å structure of the virus’s polymerase has also shown how the drug remdesivir binds to it, which could help design more effective antiviral agents against COVID-19 (Science 2020, DOI: 10.1126/science.abc1560).
Others are using cryo-EM to investigate why SARS-CoV-2 spreads so efficiently. The University of Helsinki’s Sarah J. Butcher, who heads Finland’s national cryo-EM facility, is part of a team that recently discovered how a cellular receptor called neuropilin-1 might play a key role in infection. Cells in the nasal cavity are peppered with neuropilin-1, and studies in mice, which have not yet undergone peer review, show that the virus exploits the receptor to enter cells (bioRxiv 2020, DOI: 10.1101/2020.06.07.137802). The team now plans to study the structural details of how virus proteins interact with neuropilin-1 and other proteins, which could reveal how to block the process. Butcher says that cryo-EM is ideal for these studies because it can provide clear images at widely different size scales, from specific receptor interactions to the whole virus.

Meanwhile, Carragher is involved in nine COVID-19 projects studying interactions between viral spike proteins and antibodies with an eye toward vaccine development. Several more projects target interactions between candidate drugs and the virus’s polymerase. One collaboration, published on a preprint server, has already identified several monoclonal antibodies that may be promising therapeutic agents (bioRxiv 2020, DOI: 10.1101/2020.06.17.153486).
“Cryo-EM is providing unique insights into the biology of SARS-CoV-2,” Scheres says. “Some of these targets are impossible with X-ray crystallography.”
Voltage drop
Despite this catalog of successes, Russo says that cryo-EM is still being held back by its high cost. He is working with Nobel Prize winner Henderson to make a smaller microscope that can deliver good performance at a fraction of the price. The team’s strategy for lowering cost is to reduce the instrument’s operating voltage.
Today’s commercial microscopes typically fire electrons at about 300 keV—the highest practical voltage for these instruments—which gives the particles enough momentum to provide high-quality data as they scatter off the protein’s atoms.
The convoluted evolution of the various microscope components also steered instrument makers toward that particular voltage. But was 300 keV really the best choice? Russo and his colleagues went back to the drawing board to figure out how to get the optimum amount of useful electron scattering off a protein’s atoms, while minimizing radiation damage. “It turns out the optimum voltage for all of the single-particle structure work is around 100 keV, not 300 keV like we’ve been using all this time,” Russo says. “This was a big shock.”
Advertisement
Russo’s team then built a 100 keV cryo-EM machine from spare parts and used it to acquire five structures in a week—similar to what a commercial machine could manage—with resolutions as good as 3.4 Å (IUCrJ 2019, DOI: 10.1107/S2052252519012612). The 100 keV microscope is smaller, has lower running costs, and needs less safety equipment than conventional microscopes.
Russo had hoped to persuade microscope manufacturers to make a 100 keV cryo-EM system suitable for workhorse protein structure determination, but so far he has been unsuccessful. “They’re selling Rolls-Royces, and we want Ford Focuses,” he says. So his team is building its own prototype 100 keV microscope with new parts optimized for that voltage, which he hopes will match the performance of today’s premier machines. With a price tag under $1 million, it could open up cryo-EM to thousands more researchers, he says.
And he remains hopeful that microscope manufacturers will come around to his lower-voltage system—after all, “you can sell a lot more Ford Focuses than you can Rolls-Royces.”

Mark Peplow is a freelance writer. A version of this story first appeared in ACS Central Science: cenm.ag/cryoresolution.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter