Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Nanomaterials
Adeyemi Adeleye
This environmental chemist develops nanomaterials to save contaminated soil and water
by Katherine Bourzac
July 15, 2022
| A version of this story appeared in
Volume 100, Issue 25

Credit: Will Ludwig/C&EN/Tim Peacock/Ziwei Han (Research); Yang H. Ku/C&EN/Shutterstock (Illustration)
Adeyemi Adeleye struggles with his houseplant relationships. During a video call, he holds his office plant up to the camera. Some of the once-green leaves have gone brown, and he’s worried. “I feel that it just doesn’t look as happy,” he says. “This is my cycle with plants. We start on a good note, at some point something goes wrong, and the relationship nosedives.”
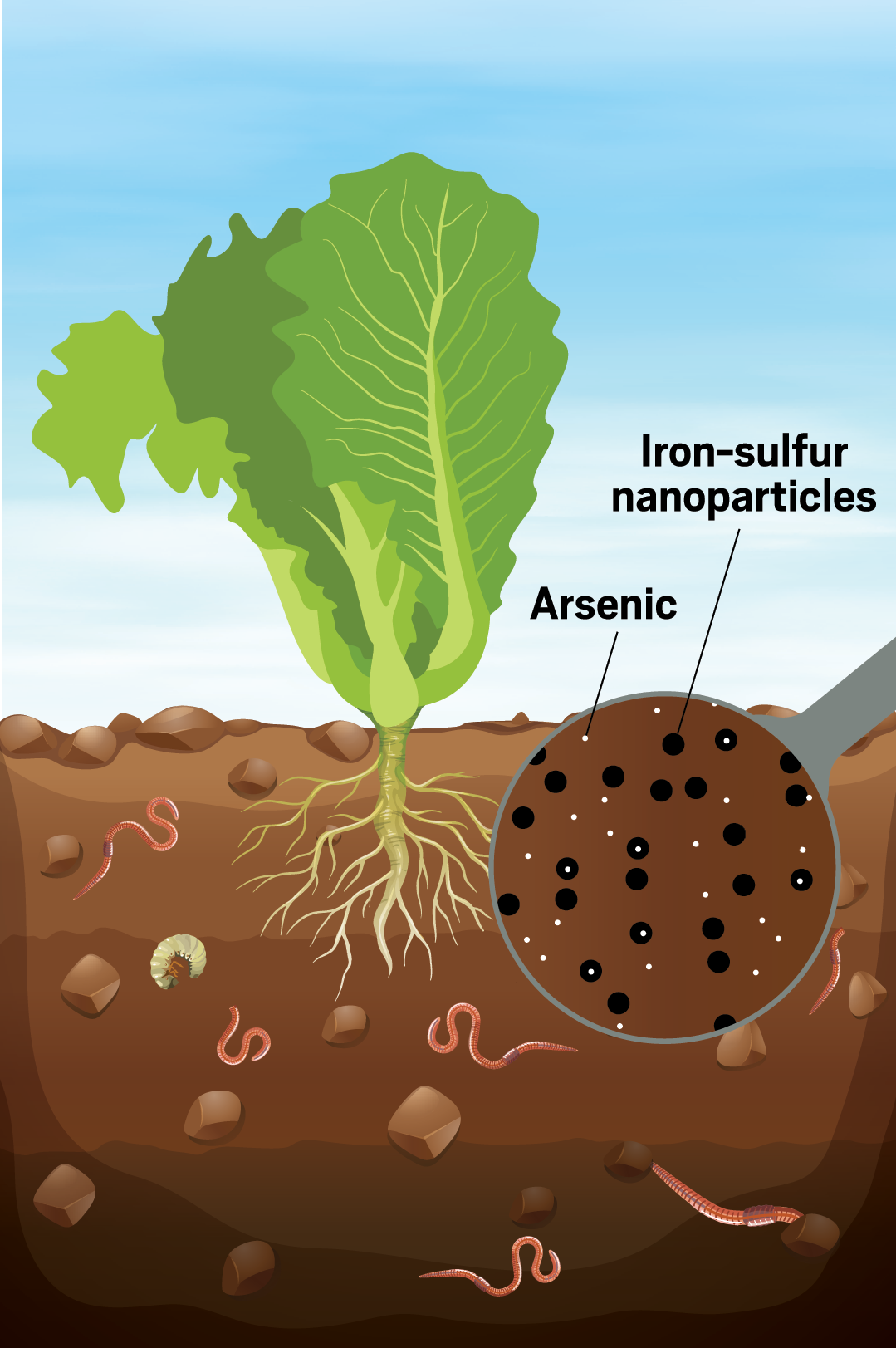
Luckily, Adeleye, an environmental chemist at the University of California, Irvine, is better at building relationships with collaborators from different disciplines. His work on nanoparticles for environmental remediation and water treatment spans chemistry, materials science, biology, and these days, agriculture. Adeleye designs nanomaterials to trap toxic arsenic in contaminated soil without harming critters such as earthworms and plants like lettuce.
Advertisement
Exposure to arsenic, typically from drinking contaminated water or eating crops like rice that take it up from the soil, causes heart disease and cancer. The metalloid is naturally present in some parts of the world, including the US and Bangladesh, and is stirred up by agricultural and mining activities. Arsenic has also been used in pesticides, but most of these products have been phased out as the human health impacts of arsenic have become widely known. Dealing with arsenic contamination is extremely challenging, Adeleye says, because the metalloid can’t be broken down like organic contaminants. He hopes to immobilize arsenic within contaminated soils so that it won’t wash into bodies of water during rainstorms and so that crops might one day be safely grown on contaminated land.
Iron nanoparticles show promise for immobilizing arsenic in the lab—they have a high surface area and readily reduce and immobilize the metalloid—but these particles can be toxic to living creatures. “We’ve had a lot of scientific and technological breakthroughs that later on we figured out, oops, this was a bad idea,” Adeleye says. His recent work shows that nanoparticles made up of a mix of iron and sulfur are nearly as effective at immobilizing arsenic as those made from elemental iron while being less toxic to earthworms.
The next question, Adeleye says, is, “Hey, can we actually reuse that soil for growing things?” For example, he wonders if these nanoparticles could be used in urban gardens where city dwellers grow food. Adeleye and green-thumbed collaborators are studying how lettuce, which tends to take up toxic metals and arsenic, grows in contaminated soil treated with the nanoparticles. They’re still analyzing the results, but Adeleye’s nanotechnological soil treatment seems to have a protective effect. Lettuce grown in arsenic-contaminated soil is stunted, but lettuce grown in contaminated soil mixed with the nanoparticles grows more normally. Whether the lettuce is safe to eat will require further study.
Not only is Adeleye a skilled scientist, but he’s also great at building a team, says Arturo Keller, an environmental scientist at the University of California, Santa Barbara. Keller, who was Adeleye’s adviser through graduate school and a postdoc, says Adeleye got more done by recruiting a team of undergrads to work on his projects. “He was really good at making sure they understood the work” and were listed as authors on the resulting publications, Keller says. “He makes sure everyone is feeling comfortable.”
Adeleye is now using artificial intelligence to design tailored versions of the nanoparticles—for example, predicting ways to make them interact with arsenic more strongly while decreasing their potential toxicity. He’s also exploring their use for wastewater treatment, including removing and breaking down per- and polyfluoroalkyl substances (PFAS), which are toxic, persistent organic pollutants commonly found in the water supply.
All that and fretting about his office plant. “I’m still hopeful this one will live and be happy,” he says.
Vitals
Current affiliation: University of California, Irvine
Age: 39
PhD alma mater: University of California, Santa Barbara
Hometown: Lagos, Nigeria
If I were an element, I'd be: “Iron. It is abundant and it has multiple oxidation states, which allows it to be essential and versatile. I strive to be versatile and multidisciplinary in my approach to solving scientific problems.”
My alternate-universe career: “There is a chance I would have gone into acting.”
See the Talented 12 present their work at a virtual symposium Sept. 19, 20, and 21. Register for free at cenm.ag/t12symposium.

|

|
Adeyemi Adeleye struggles with his houseplant relationships. During a video call, he holds his office plant up to the camera. Some of the once-green leaves have gone brown, and he’s worried. “I feel that it just doesn’t look as happy,” he says. “This is my cycle with plants. We start on a good note, at some point something goes wrong, and the relationship nosedives.”<
Vitals
▸ Current affiliation: University of California, Irvine
▸ Age: 39
▸ PhD alma mater: University of California, Santa Barbara
▸ Hometown: Lagos, Nigeria
▸ If I were an element, I’d be: “Iron. It is abundant and it has multiple oxidation states, which allows it to be essential and versatile. I strive to be versatile and multidisciplinary in my approach to solving scientific problems.”
▸ My alternate-universe career: “There is a chance I would have gone into acting.”
Luckily, Adeleye, an environmental chemist at the University of California, Irvine, is better at building relationships with collaborators from different disciplines. His work on nanoparticles for environmental remediation and water treatment spans chemistry, materials science, biology, and these days, agriculture. Adeleye designs nanomaterials to trap toxic arsenic in contaminated soil without harming critters such as earthworms and plants like lettuce.
Exposure to arsenic, typically from drinking contaminated water or eating crops like rice that take it up from the soil, causes heart disease and cancer. The metalloid is naturally present in some parts of the world, including the US and Bangladesh, and is stirred up by agricultural and mining activities. Arsenic has also been used in pesticides, but most of these products have been phased out as the human health impacts of arsenic have become widely known. Dealing with arsenic contamination is extremely challenging, Adeleye says, because the metalloid can’t be broken down like organic contaminants. He hopes to immobilize arsenic within contaminated soils so that it won’t wash into bodies of water during rainstorms and so that crops might one day be safely grown on contaminated land.
Iron nanoparticles show promise for immobilizing arsenic in the lab—they have a high surface area and readily reduce and immobilize the metalloid—but these particles can be toxic to living creatures. “We’ve had a lot of scientific and technological breakthroughs that later on we figured out, oops, this was a bad idea,” Adeleye says. His recent work shows that nanoparticles made up of a mix of iron and sulfur are nearly as effective at immobilizing arsenic as those made from elemental iron while being less toxic to earthworms.
The next question, Adeleye says, is, “Hey, can we actually reuse that soil for growing things?” For example, he wonders if these nanoparticles could be used in urban gardens where city dwellers grow food. Adeleye and green-thumbed collaborators are studying how lettuce, which tends to take up toxic metals and arsenic, grows in contaminated soil treated with the nanoparticles. They’re still analyzing the results, but Adeleye’s nanotechnological soil treatment seems to have a protective effect. Lettuce grown in arsenic-contaminated soil is stunted, but lettuce grown in contaminated soil mixed with the nanoparticles grows more normally. Whether the lettuce is safe to eat will require further study.
Not only is Adeleye a skilled scientist, but he’s also great at building a team, says Arturo Keller, an environmental scientist at the University of California, Santa Barbara. Keller, who was Adeleye’s adviser through graduate school and a postdoc, says Adeleye got more done by recruiting a team of undergrads to work on his projects. “He was really good at making sure they understood the work” and were listed as authors on the resulting publications, Keller says. “He makes sure everyone is feeling comfortable.”
Adeleye is now using artificial intelligence to design tailored versions of the nanoparticles—for example, predicting ways to make them interact with arsenic more strongly while decreasing their potential toxicity. He’s also exploring their use for wastewater treatment, including removing and breaking down per- and polyfluoroalkyl substances (PFAS), which are toxic, persistent organic pollutants commonly found in the water supply.
All that and fretting about his office plant. “I’m still hopeful this one will live and be happy,” he says.





















Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter