Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Computational Chemistry
Gabe Gomes
This machine learning expert trains computers to develop an intuition for chemical reactions
by Ariana Remmel
July 15, 2022
| A version of this story appeared in
Volume 100, Issue 25

Credit: Will Ludwig/C&EN/Tim Peacock/David Cochran
During one of Gabe Gomes’s first chemistry lectures at the Federal University of Rio de Janeiro, a professor walked up to the board and wrote the Schrödinger equation for the class. “All of chemistry is in this equation,” the professor said. “All you need to do is solve it.”
Gomes loved the class because he learned that quantum mechanics, like the Schrödinger equation, allows chemists to understand how a molecule’s structure and energetics can affect its behavior. That class set him on a journey of trying to reveal how molecules do what they do as 3D quantum objects.
Advertisement
Today, Gomes and his lab teach computers the fundamental principles of quantum mechanics to automate the design of new catalysts and chemical reactions. They hope these methods can speed up the discovery of innovative chemistries for building molecules.
Throughout Gomes’s career, he’s used his knack for visualizing molecules’ dynamic behavior to undertake a wide variety of problems across physical chemistry and organic synthesis. As a graduate student at Florida State University, Gomes dived headfirst into stereoelectronic effects, an approach that uses electronic configurations to describe molecules’ reactivity. Then, as a postdoc at the University of Toronto, Gomes started using artificial intelligence and machine learning to predict chemical properties from the structures of small molecules like organophosphate ligands.
Now with his own lab at Carnegie Mellon University, Gomes has turned to catalyst design. When chemists want to develop new catalysts, they normally tweak an existing catalyst’s structure until they achieve the desired reactivity in the lab. But this process can be slow and inefficient, says Igor Alabugin, who was Gomes’s graduate adviser at Florida State University. Gomes and his lab want to flip that process on its head.
He aims to impart computers with a kind of chemical intuition that could allow algorithms to propose new functional catalysts from scratch. In this inverse design framework, “you tell me the properties that you need in your next material, and I’ll give you the structure,” Gomes says. To build programs with this know-how, he and his lab are training computers to handle chemicals as 3D quantum objects. By stripping molecules down to their fundamental physical properties, Gomes hopes machines can engineer novel chemistry from the ground up.
It’s an ambitious project that could have far-reaching applications, including discovering new reactions in green chemistry and designing the next generation of climate-resilient materials, Alabugin says. He credits Gomes’s intellectual bravery and contagious curiosity for his success. “It was immediately obvious that he is very talented,” Alabugin says. “He’s always trying to learn something new, to solve a new problem.”
Despite Gomes’s computational prowess, he resists being labeled as a computational chemist. “As the central science, we shouldn’t be thinking of ourselves with those divisions for much longer,” he says. Gomes is looking toward an exciting future for chemists propelled by the power of chemically savvy computers. “Can machines dream of molecules that cure cancer? I want you to say yes, but the reality is that we are learning,” Gomes says.

Vitals
Current affiliation: Carnegie Mellon University
Age: 33
PhD alma mater: Florida State University
Hometown: Rio de Janeiro
If I were an element, I'd be: “Carbon. At the end of the day, the sheer versatility of molecules that this one element is involved in makes me think that we have something in common.”
My alternate-universe career: “Something related to music and computers. I used to think I’d own a studio and produce bands and artists, help them find their sound.”
See the Talented 12 present their work at a virtual symposium Sept. 19, 20, and 21. Register for free at cenm.ag/t12symposium.

|
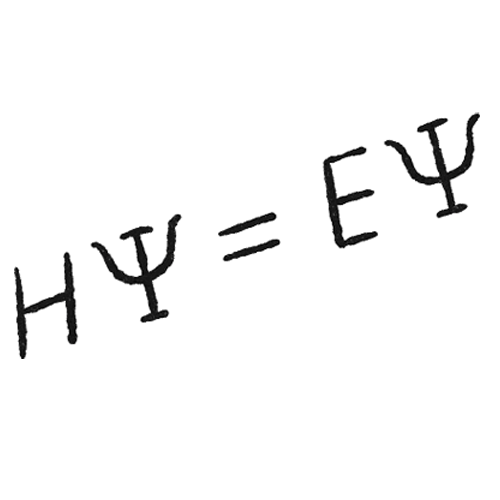
|
During one of Gabe Gomes’s first chemistry lectures at the Federal University of Rio de Janeiro, a professor walked up to the board and wrote the Schrödinger equation for the class. “All of chemistry is in this equation,” the professor said. “All you need to do is solve it.”
Vitals
▸ Current affiliation: Carnegie Mellon University
▸ Age: 33
▸ PhD alma mater: Florida State University
▸ Hometown: Rio de Janeiro
▸ If I were an element, I’d be: “Carbon. At the end of the day, the sheer versatility of molecules that this one element is involved in makes me think that we have something in common.”
▸ My alternate-universe career: “Something related to music and computers. I used to think I’d own a studio and produce bands and artists, help them find their sound.”
Gomes loved the class because he learned that quantum mechanics, like the Schrödinger equation, allows chemists to understand how a molecule’s structure and energetics can affect its behavior. That class set him on a journey of trying to reveal how molecules do what they do as 3D quantum objects.
Today, Gomes and his lab teach computers the fundamental principles of quantum mechanics to automate the design of new catalysts and chemical reactions. They hope these methods can speed up the discovery of innovative chemistries for building molecules.
Throughout Gomes’s career, he’s used his knack for visualizing molecules’ dynamic behavior to undertake a wide variety of problems across physical chemistry and organic synthesis. As a graduate student at Florida State University, Gomes dived headfirst into stereoelectronic effects, an approach that uses electronic configurations to describe molecules’ reactivity. Then, as a postdoc at the University of Toronto, Gomes started using artificial intelligence and machine learning to predict chemical properties from the structures of small molecules like organophosphate ligands.
Now with his own lab at Carnegie Mellon University, Gomes has turned to catalyst design. When chemists want to develop new catalysts, they normally tweak an existing catalyst’s structure until they achieve the desired reactivity in the lab. But this process can be slow and inefficient, says Igor Alabugin, who was Gomes’s graduate adviser at Florida State University. Gomes and his lab want to flip that process on its head.
He aims to impart computers with a kind of chemical intuition that could allow algorithms to propose new functional catalysts from scratch. In this inverse design framework, “you tell me the properties that you need in your next material, and I’ll give you the structure,” Gomes says. To build programs with this know-how, he and his lab are training computers to handle chemicals as 3D quantum objects. By stripping molecules down to their fundamental physical properties, Gomes hopes machines can engineer novel chemistry from the ground up.
It’s an ambitious project that could have far-reaching applications, including discovering new reactions in green chemistry and designing the next generation of climate-resilient materials, Alabugin says. He credits Gomes’s intellectual bravery and contagious curiosity for his success. “It was immediately obvious that he is very talented,” Alabugin says. “He’s always trying to learn something new, to solve a new problem.”
Despite Gomes’s computational prowess, he resists being labeled as a computational chemist. “As the central science, we shouldn’t be thinking of ourselves with those divisions for much longer,” he says. Gomes is looking toward an exciting future for chemists propelled by the power of chemically savvy computers. “Can machines dream of molecules that cure cancer? I want you to say yes, but the reality is that we are learning,” Gomes says.




















Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter