Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Green Chemistry
Connora Technologies
A first-ever route to recyclable composites
by Alexander H. Tullo
November 2, 2015
| A version of this story appeared in
Volume 93, Issue 43
Lightweight carbon fiber composites are making our automobiles and airplanes stronger and more fuel efficient. But ideally, we would reuse these valuable materials when cars and planes are scrapped.
Connora Technologies
Launched: 2012
Location: Hayward, Calif.
Focus: Composite resins for transportation and sporting goods
Technology: Epoxy hardeners that make composites recyclable
Founders: Rey Banatao and Stefan J. Pastine
Funding: Entropy Research Labs, Chemical Angel Network, and Samsung Ventures
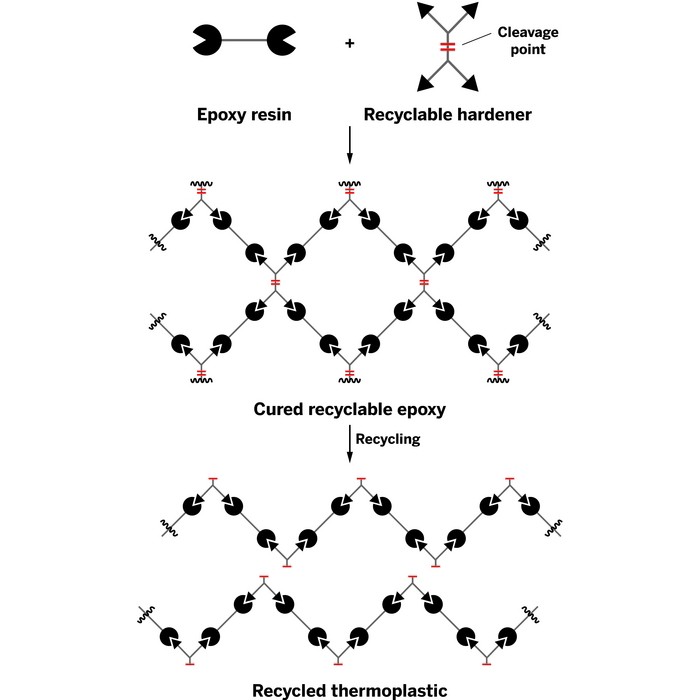
There’s no practical way to do this yet. Carbon fiber composites are held together by thermoset resins, usually epoxies, which aren’t recyclable like thermoplastics are. Thermoplastics can be remelted and molded again. Thermosets, when heated, break apart long before they would ever melt.
Connora Technologies wants to change that by programming recyclability into thermosets before they are used the first time. The founders hope that the technology will enable greater use of lightweight composites in applications such as cars.
To make epoxy resins, epichlorohydrin is reacted with bisphenol A to form bisphenol A diglycidyl ether, a viscous liquid. The ether is mixed with curing agents, which have amine groups that react with the ether’s epoxide groups, cross-linking the molecules into a hard, three-dimensional network of covalently bonded material.
Connora has developed a new class of curing agents it calls Recyclamine. These polyamino acetal-based molecules have built-in acid-cleavable bonds that become part of the epoxy structure.
Stefan J. Pastine, Connora’s chief technology officer, got the idea while a postdoc in Jean M. J. Fréchet’s group at the University of California, Berkeley. The group works with responsive materials—molecules, such as photoresists, that change under specific conditions. Looking for an area within polymers to apply the idea, Pastine hit upon epoxies. “Here’s this material that is not recyclable, and the use of composites was starting to increase,” he recalls.
Connora is building a recycling system around Recyclamine. In it, carbon fiber composites are immersed in an acetic acid bath at about 100 °C. In less than two hours, all the cleavable bonds break and the resin separates from the carbon fiber. The clean fiber is filtered out. Neutralization of the acid precipitates the dissolved polymer out of the solution.
The reclaimed polymer is a thermoplastic, not an epoxy that can be cross-linked into a thermoset again. According to Rey Banatao, Connora’s CEO, the polymer has physical properties similar to nylon and polypropylene. Being derived from an epoxy, the polymer also has adhesive properties.
But the more compelling target of the recycling process may be the carbon fiber. “Thermoplastics are cheap,” Banatao says. “Carbon fiber is expensive.”
Other methods to recycle carbon fiber rely on pyrolysis—burning the epoxy off. However, Banatao says pyrolysis can degrade the fibers, reducing their value and performance. He claims the carbon fibers that come out of Connora’s process are pristine.
Connora is working with an unnamed chemical manufacturer, which is making several tons of Recyclamine for the company’s commercial launch this year. For its first customers, Connora is looking to makers of sporting goods such as bicycle frames, skis, skateboards, and surfboards.
The company hopes to eventually supply Recyclamine to the auto industry. Regulations in Europe mandate that recyclable materials make up 85% of each vehicle, and Banatao contends that Recyclamine will help composite materials meet those requirements. The process also could allow automakers to recycle waste generated while making parts from composites.
Connora has received funding through the Entropy Research Labs, Chemical Angel Network, and Samsung Ventures. The company is now pursuing a Series A round of venture capital funding.
Banatao sees a day when Connora’s materials have a big impact. “It is basically the first example of being able to convert a thermoset into a thermoplastic that could be industrially viable,” he says.
Find out what Connora Technologies has been up to since it appeared on C&EN’s 2015 Top Start-ups List by clicking here.






Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter