Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Drug Discovery
Lynn McGregor
Drug-discovery dynamo is using chemical biology to rewrite the rules of small molecules
by Lisa M. Jarvis
August 14, 2020
| A version of this story appeared in
Volume 98, Issue 31

Credit: Courtesy of Lynn McGregor (McGregor); Novartis (E3 ligase); RCSB PDB (KRAS); Courtesy of Colin Garvie (protein gels)
The human proteome is vast and, to the chagrin of drug developers, largely out of reach. Of the tens of thousands of proteins that perform our cells’ various functions, only about 15% can be accessed with conventional small molecules and antibody drugs.
Chemical biologist Lynn McGregor is helping to change those stats. Throughout her academic training and nascent career in industry, she has developed expertise in a range of technologies that could help open up the possibilities of the proteome.
Advertisement
As a postdoctoral fellow in Kevan Shokat’s lab at the University of California, San Francisco, McGregor took on one of the most daunting drug targets out there, KRAS. The protein is notorious for its frequent involvement in cancer—and for the decades drug developers have spent trying to crack it.
Shokat’s lab had already succeeded in finding the first tool compound to block a mutant form of KRAS called G12C. McGregor was tasked with blocking another KRAS variant, G12D.
In the end, she didn’t succeed (though it’s worth noting that to date no one has been able to develop a KRAS G12D inhibitor). “There’s probably thousands of people with a failed RAS project, and I’m one of them,” McGregor jokes.
But just tackling such a challenging endeavor shows McGregor’s mettle, Shokat says. “It takes a special scientist willing to take on these kinds of projects because starting out you know the odds are against success.”
McGregor has continued to explore new and difficult frontiers of drug discovery. After her postdoc, she took a job in Novartis’s chemical biology group, where one of her projects has been to improve the design of protein degraders—small-molecule drugs that break down, rather than simply block the activity of proteins. These bifunctional small molecules feature one end that binds to a protein of interest and another end that binds to an E3 ligase, which is responsible for recruiting the other players in the cell’s protein disposal system.
Researchers have spent the past several years trying to work out the rules for constructing these complex molecules, which to date have largely been fashioned through trial and error. McGregor, along with Novartis colleagues and their partners at the University of California, Berkeley, is helping develop a more rational approach.
The collaborators recently tethered three types of E3 ligases to the blood cancer treatment dasatinib to understand how each affects the breakdown of a fusion protein, BCR-ABL. It turns out that the protein degrader featuring the natural product nimbolide was highly specific for that cancer-driver, while the two other E3 ligases either were inactive or didn’t break down the entire complex.
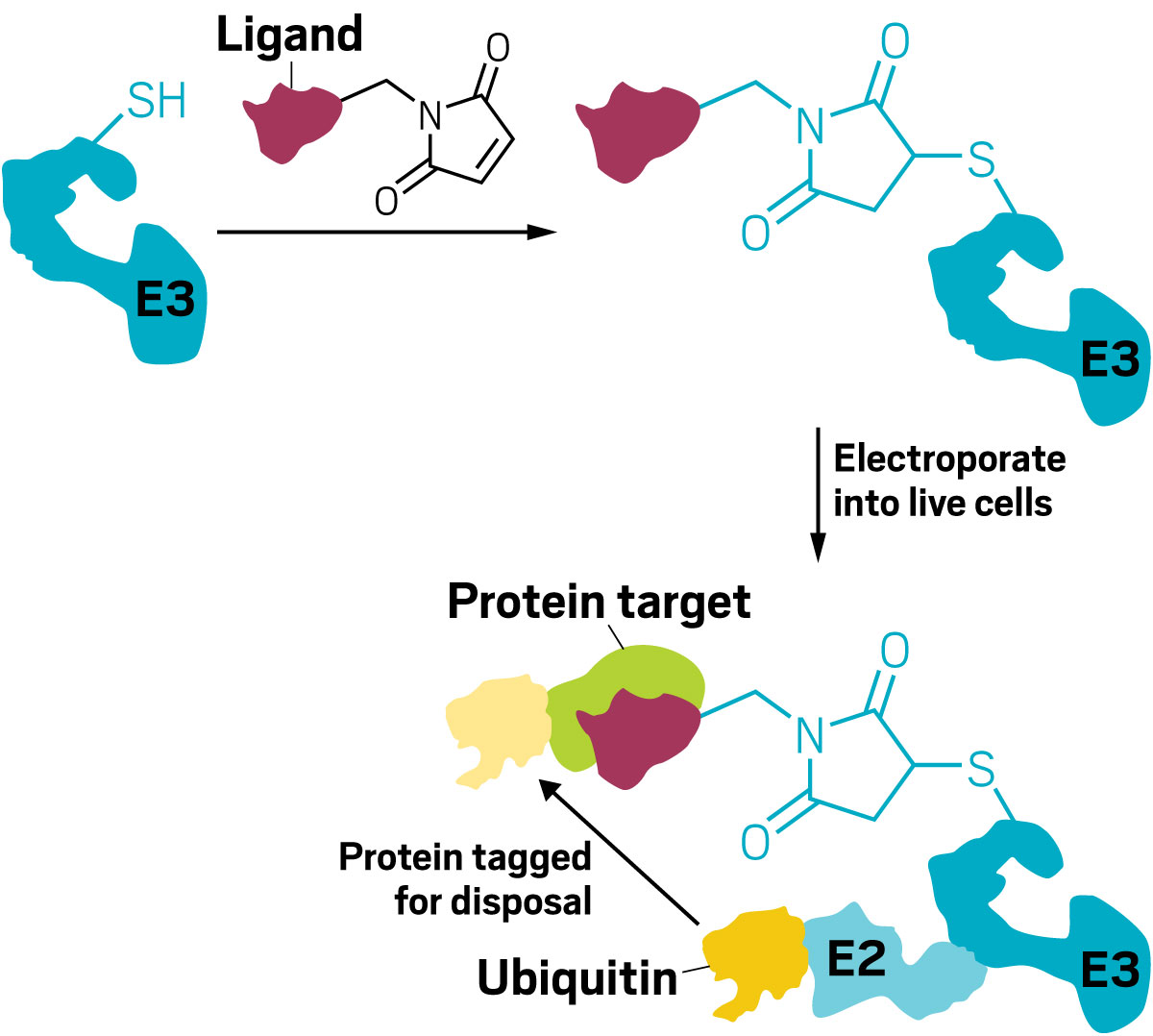
Given those differences, McGregor then began exploring easier methods for choosing the right E3 ligase. So far the field has focused on only a few of the hundreds of E3 ligases available, and Novartis scientists wanted an easy filter for the best ligase for a particular protein. In soon-to-be-published work, McGregor and a small team of interns from Dana-Farber Cancer Institute came up with a method that uses simple chemistry to directly modify an E3 ligase and put it back into cells to ask whether it causes a protein of interest to be degraded.
It’s this type of work, accomplished with many different partners, that has made her stand out within a big company like Novartis. “Lynn’s humble and collaborative nature provides balance with her steely determination to advance chemical biology at Novartis,” says John Tallarico, head of chemical biology at Novartis Institutes for BioMedical Research. “Her talents include not only scientific acumen but that special people ability to bring experts from different disciplines together at a high level to get important questions answered.”
Vitals
Current affiliation: Novartis
Age: 34
PhD alma mater: Harvard University
Hometown: Guilford, Connecticut
If I weren’t a chemist, I’d be: A sustainability scientist. “Both drug discovery and sustainability have the goal of making sure more people can live long, healthy lives.”
If I were an element, I’d be: Sodium. “Despite my best efforts, I can be a bit prone to salty language and the rare fiery outburst.”
The human proteome is vast and, to the chagrin of drug developers, largely out of reach. Of the tens of thousands of proteins that perform our cells’ various functions, only about 15% can be accessed with conventional small molecules and antibody drugs.
Vitals
▸ Current affiliation: Novartis
▸ Age: 34
▸ PhD alma mater: Harvard University
▸ Hometown: Guilford, Connecticut
▸ If I weren’t a chemist, I’d be: A sustainability scientist. “Both drug discovery and sustainability have the goal of making sure more people can live long, healthy lives.”
▸ If I were an element, I’d be: Sodium. “Despite my best efforts, I can be a bit prone to salty language and the rare fiery outburst.”
Chemical biologist Lynn McGregor is helping to change those stats. Throughout her academic training and nascent career in industry, she has developed expertise in a range of technologies that could help open up the possibilities of the proteome.
As a postdoctoral fellow in Kevan Shokat’s lab at the University of California, San Francisco, McGregor took on one of the most daunting drug targets out there, KRAS. The protein is notorious for its frequent involvement in cancer—and for the decades drug developers have spent trying to crack it.
Shokat’s lab had already succeeded in finding the first tool compound to block a mutant form of KRAS called G12C. McGregor was tasked with blocking another KRAS variant, G12D.
In the end, she didn’t succeed (though it’s worth noting that to date no one has been able to develop a KRAS G12D inhibitor). “There’s probably thousands of people with a failed RAS project, and I’m one of them,” McGregor jokes.
But just tackling such a challenging endeavor shows McGregor’s mettle, Shokat says. “It takes a special scientist willing to take on these kinds of projects because starting out you know the odds are against success.”
McGregor has continued to explore new and difficult frontiers of drug discovery. After her postdoc, she took a job in Novartis’s chemical biology group, where one of her projects has been to improve the design of protein degraders—small-molecule drugs that break down, rather than simply block the activity of proteins. These bifunctional small molecules feature one end that binds to a protein of interest and another end that binds to an E3 ligase, which is responsible for recruiting the other players in the cell’s protein disposal system.
Researchers have spent the past several years trying to work out the rules for constructing these complex molecules, which to date have largely been fashioned through trial and error. McGregor, along with Novartis colleagues and their partners at the University of California, Berkeley, is helping develop a more rational approach.
The collaborators recently tethered three types of E3 ligases to the blood cancer treatment dasatinib to understand how each affects the breakdown of a fusion protein, BCR-ABL. It turns out that the protein degrader featuring the natural product nimbolide was highly specific for that cancer-driver, while the two other E3 ligases either were inactive or didn’t break down the entire complex.
Given those differences, McGregor then began exploring easier methods for choosing the right E3 ligase. So far the field has focused on only a few of the hundreds of E3 ligases available, and Novartis scientists wanted an easy filter for the best ligase for a particular protein. In soon-to-be-published work, McGregor and a small team of interns from Dana-Farber Cancer Institute came up with a method that uses simple chemistry to directly modify an E3 ligase and put it back into cells to ask whether it causes a protein of interest to be degraded.
It’s this type of work, accomplished with many different partners, that has made her stand out within a big company like Novartis. “Lynn’s humble and collaborative nature provides balance with her steely determination to advance chemical biology at Novartis,” says John Tallarico, head of chemical biology at Novartis Institutes for BioMedical Research. “Her talents include not only scientific acumen but that special people ability to bring experts from different disciplines together at a high level to get important questions answered.”




















Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter