Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Josep Cornella
Catalysis connoisseur is developing reagents that make chemistry faster and easier
by Bethany Halford
August 14, 2020
| A version of this story appeared in
Volume 98, Issue 31

Credit: Carla Obradors (Cornella); Courtesy of Josep Cornella (structures); Shutterstock (bismuth)
It might be overstating things to call bis(1,5-cyclooctadiene)nickel (also known as Ni(COD)2) Josep Cornella’s nemesis. But Cornella, a group leader at the Max Planck Institute for Kohlenforschung, readily admits that he was never fond of the popular but air-sensitive coupling catalyst.
He recalls his time as a graduate student and a postdoctoral fellow working with the reagent with antipathy, overheated and sweating while he awkwardly reached into a glove box. Sure, Ni(COD)2 works well, he says, but it was also the only option for chemists who want to work with nickel(0). “In the back of my mind I thought, ‘If somebody makes an air-stable nickel(0) complex, that’s going to be, for all the chemists that use nickel, so helpful,’ ” Cornella says. But he also thought it would be a tough project, and left it at that.
Advertisement
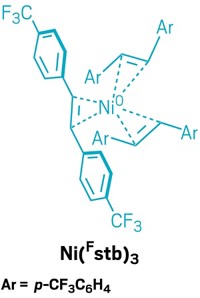
Then, one day in 2019, Cornella’s graduate student, who was working on an unrelated project, walked into his office with a vial containing a nickel(0) complex that seemed to be stable in air. Cornella was stunned. His group tweaked this molecule to create nickel(0) surrounded by three stilbene ligands that bear trifluoromethyl groups. The bulky ligands protect the metal from oxygen, making the complex stable for days at room temperature and months in the freezer. It can be used to generate active catalysts for more than a dozen reactions, and Cornella is currently in negotiations with a reagent company to make the patented catalyst available for chemists to buy.
As a catalysis specialist, Cornella says he’s interested in making reagents that give chemists better, faster, and more efficient reactions. But as his lab explores these catalysts, there’s no long-term agenda. “We are mostly driven by curiosity,” he says.
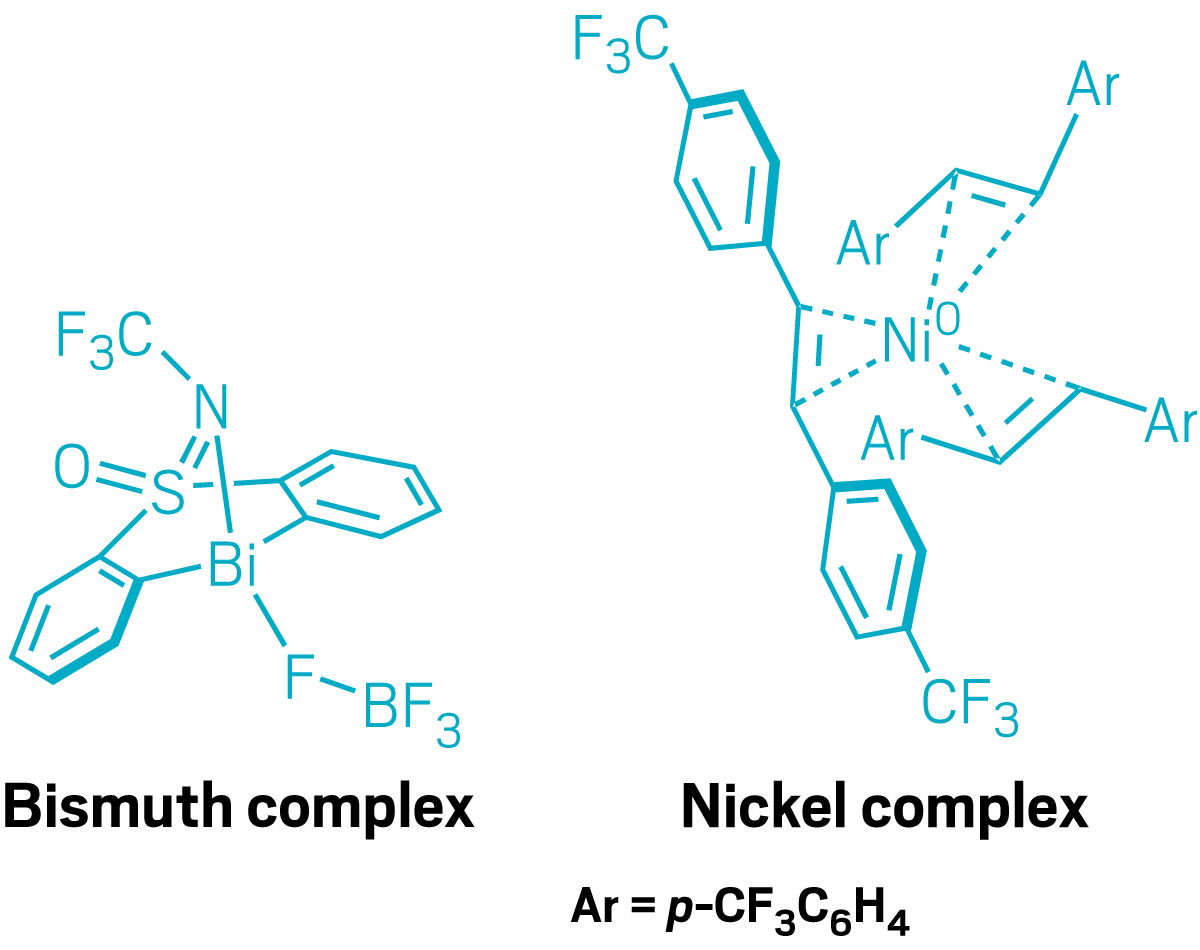
For example, Cornella wondered if it was possible to make bismuth behave like a transition metal. Bismuth is generally considered benign, but its nearest neighbors—lead, tin, antimony, tellurium, and polonium—can all be poisons. This makes bismuth appealing for anyone looking to make the most of heavy elements’ special properties, such as their ability to coordinate many ligands (bismuth can handle up to 9) and adopt unusual geometries. Cornella’s group fashioned a bismuth catalyst that fluorinates aryl boronic acids and esters, a transformation commonly reserved for transition-metal catalysts.
Cornella’s path to a career in chemistry wasn’t always clear. He finished his master’s degree at the University of Barcelona in 2008, just as the global recession was hitting Spain. “Fellowships stopped. Everything stopped. I wanted to do a PhD, but there was no money,” he recalls. Although he spoke very little English, Cornella applied to do his doctoral work with Igor Larrosa, who was at Queen Mary University of London at the time. “He realized that I understood the chemistry and he told me that the language would come in a couple of months,” Cornella says.
A Marie Skłodowska-Curie Individual Fellowship brought Cornella back to Spain, where he studied with Ruben Martin at the Institute of Chemical Research of Catalonia. He then did a second postdoc with Phil Baran of Scripps Research in California before taking his current position in Germany.
Baran calls Cornella’s research strikingly original and bold. “Rather than chasing fashions and low-hanging fruit he is spearheading inspiring directions that are not only wholly unique but also have immediate practical utility and impact,” Baran says.
Cornella says he’s just doing something that he loves. “Be passionate about what you do,” he says. “If you’re not passionate, you will quit very quickly.”
Vitals
Current affiliation: Max Planck Institute for Kohlenforschung
Age: 35
PhD alma mater: Queen Mary University of London
Hometown: La Bisbal del Penedès, Spain
If I weren’t a chemist, I’d be: A graphic designer or an architect. “I like the beauty of slick designs, simple but powerful.”
If I were an element, I’d be: Hydrogen. “The important thing is that I could be anywhere in the universe.”
It might be overstating things to call bis(1,5-cyclooctadiene)nickel (also known as Ni(COD)2) Josep Cornella’s nemesis. But Cornella, a group leader at the Max Planck Institute for Kohlenforschung, readily admits that he was never fond of the popular but air-sensitive coupling catalyst.
Vitals
▸ Current affiliation: Max Planck Institute for Kohlenforschung
▸ Age: 35
▸ PhD alma mater: Queen Mary University of London
▸ Hometown: La Bisbal del Penedès, Spain
▸ If I weren’t a chemist, I’d be: A graphic designer or an architect. “I like the beauty of slick designs, simple but powerful.”
▸ If I were an element, I’d be: Hydrogen. “The important thing is that I could be anywhere in the universe.”
He recalls his time as a graduate student and a postdoctoral fellow working with the reagent with antipathy, overheated and sweating while he awkwardly reached into a glove box. Sure, Ni(COD)2 works well, he says, but it was also the only option for chemists who want to work with nickel(0). “In the back of my mind I thought, ‘If somebody makes an air-stable nickel(0) complex, that’s going to be, for all the chemists that use nickel, so helpful,’ ” Cornella says. But he also thought it would be a tough project, and left it at that.
Then, one day in 2019, Cornella’s graduate student, who was working on an unrelated project, walked into his office with a vial containing a nickel(0) complex that seemed to be stable in air. Cornella was stunned. His group tweaked this molecule to create nickel(0) surrounded by three stilbene ligands that bear trifluoromethyl groups. The bulky ligands protect the metal from oxygen, making the complex stable for days at room temperature and months in the freezer. It can be used to generate active catalysts for more than a dozen reactions, and Cornella is currently in negotiations with a reagent company to make the patented catalyst available for chemists to buy.
As a catalysis specialist, Cornella says he’s interested in making reagents that give chemists better, faster, and more efficient reactions. But as his lab explores these catalysts, there’s no long-term agenda. “We are mostly driven by curiosity,” he says.
For example, Cornella wondered if it was possible to make bismuth behave like a transition metal. Bismuth is generally considered benign, but its nearest neighbors—lead, tin, antimony, tellurium, and polonium—can all be poisons. This makes bismuth appealing for anyone looking to make the most of heavy elements’ special properties, such as their ability to coordinate many ligands (bismuth can handle up to 9) and adopt unusual geometries. Cornella’s group fashioned a bismuth catalyst that fluorinates aryl boronic acids and esters, a transformation commonly reserved for transition-metal catalysts.
Cornella’s path to a career in chemistry wasn’t always clear. He finished his master’s degree at the University of Barcelona in 2008, just as the global recession was hitting Spain. “Fellowships stopped. Everything stopped. I wanted to do a PhD, but there was no money,” he recalls. Although he spoke very little English, Cornella applied to do his doctoral work with Igor Larrosa, who was at Queen Mary University of London at the time. “He realized that I understood the chemistry and he told me that the language would come in a couple of months,” Cornella says.
A Marie Skłodowska-Curie Individual Fellowship brought Cornella back to Spain, where he studied with Ruben Martin at the Institute of Chemical Research of Catalonia. He then did a second postdoc with Phil Baran of Scripps Research in California before taking his current position in Germany.
Baran calls Cornella’s research strikingly original and bold. “Rather than chasing fashions and low-hanging fruit he is spearheading inspiring directions that are not only wholly unique but also have immediate practical utility and impact,” Baran says.
Cornella says he’s just doing something that he loves. “Be passionate about what you do,” he says. “If you’re not passionate, you will quit very quickly.”




















Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter